Vol 2 | Issue 1 | Jan-Apr 2016 | page:8-11 | Andrew P Smith, Catherine A Millares, Henry Cohen, William Lois.
Authers: Andrew P Smith[1], Catherine A Millares[2], Henry Cohen[2,3], William Lois[3].
[1] Department of Pharmacy, Bronx Lebanon Hospital Center, New York, United States of America.
[2] Department of Pharmacy, Touro College of Pharmacy, New York, United States of America.
[3] Department of Surgery, Kingsbrook Jewish Medical Center, New York, United States of America.
Address of Correspondence
Dr. Andrew P Smith
Department of Pharmacy Critical Care Clinical Pharmacist.
Bronx Lebanon Hospital Center, New York
United States of America.
Email: asmith@bronxleb.org
Abstract
Background: Indigotindisulfonate (Indigo Carmine, American Regent, Shirely, NY) is a blue dye that is commonly used for localizing ureteral orifices during surgery. In general, it is safe and biologically inactive, with the package insert citing only rare idiosyncratic reactions and mild pressor effects in some patients. We report a case of a severe life-threatening anaphylactoid reaction due to indigotindisulfonate following intravenous administration.
Case Report: We describe a case of a 42-year-old female admitted for a total abdominal hysterectomy. Upon arrival to the operating room, her heart rate (HR) was 80/min, blood pressure (BP) was 135/75 mm Hg, and a SpO2 of 98%. During surgery, the patient received 5 mL of slow IV bolus of 0.8 percent Indigotindisulfonate sodium injectable solution, at which time her blood pressure was 110/60 mm Hg and heart rate was 75/min. Fifteen minutes later the patient became hypotensive and bradycardic (BP = 70/50 mm Hg; HR = 37/min). The team performed advanced cardiac life support for 16 minutes, administering epinephrine 1 mg IV BOLUS x 4 doses and sodium bicarbonate 50 mEqIV BOLUS x 3 doses.
Conclusion: Although this is a rare adverse event, it is prudent to consider the possibility of these reactions when a bolus of Indigotindisulfonate is administered intravenously.
Keywords: Case report, Indigotindisulfonate Sodium, Indigo Carmine, anaphylactoid, reaction, hysterectomy.
Introduction
Indigotindisulfonate (Indigo Carmine, American Regent, Shirely, NewYork) is a blue dye that is commonly used for localizing ureteral orifices during surgery[1]. In general, it is considered safe and biologically inactive, with the manufacturer package insert citing only rare idiosyncratic reactions and mild pressor effects in some patients[2]. Severe reactions from this agent are extremely rare, with only 12 published case reports[4-8]. We report a case of cardiac arrest after an intravenous indigotindisulfonate injection during a scheduled total abdominal hysterectomy with an accompanying literature review. The patient gave written consent for authors to publish the manuscript.
Case report
A 42-yr-old, 85 kg, 160 cm, Afro-Caribbean female, American Society of Anesthesiologists physical class II, with a past medical history of uterine leiomyoma and ovarian cysts, was scheduled for a total abdominal hysterectomy. She is allergic to shellfish but has no known drug allergies.
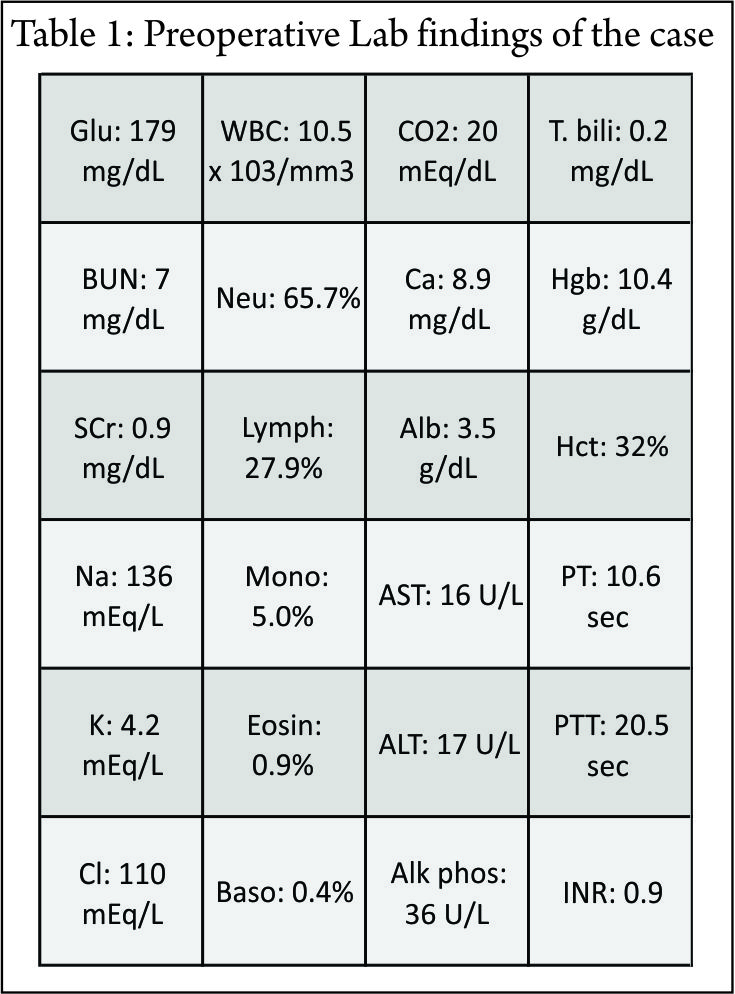
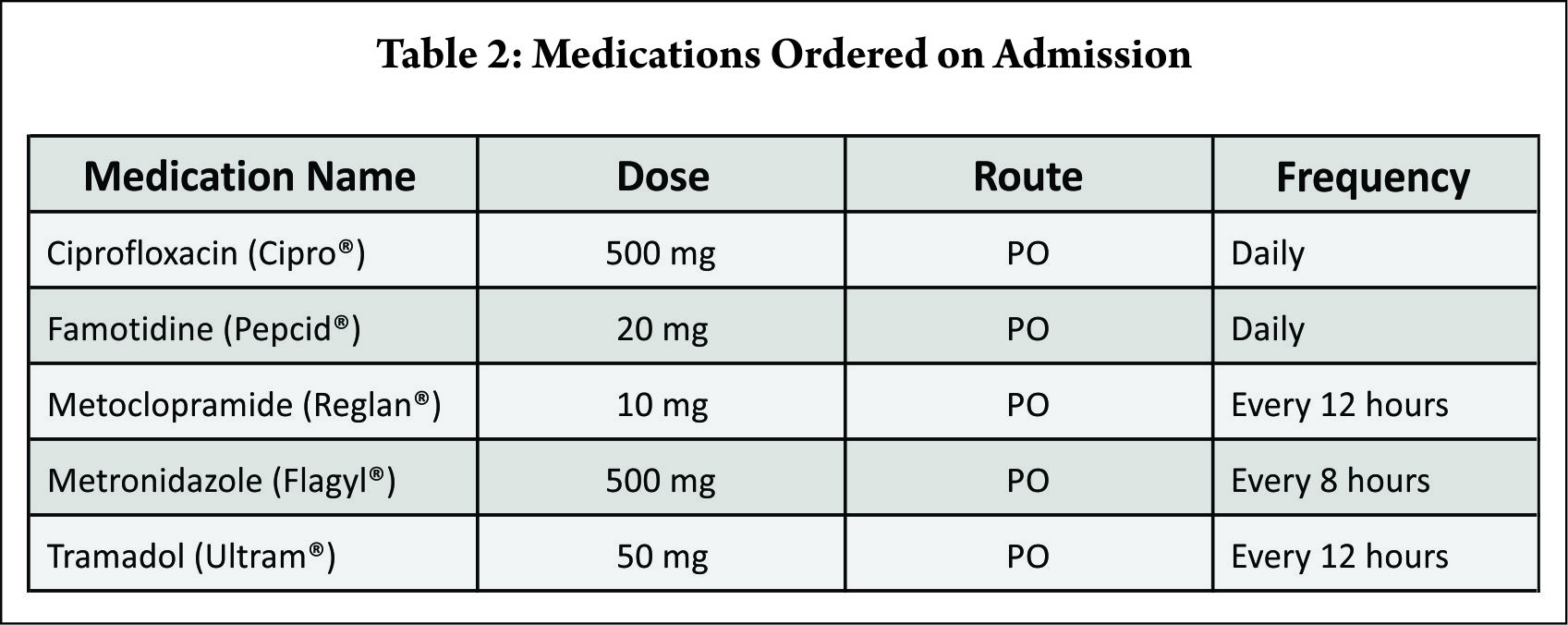
Patient denied taking prescription, over-the-counter, or herbal medications at home. Her pre-op laboratory findings were unremarkable (Table 1) and her medications on admission included ciprofloxacin, famotidine, metoclopramide, metronidazole, and tramadol (Table 2).
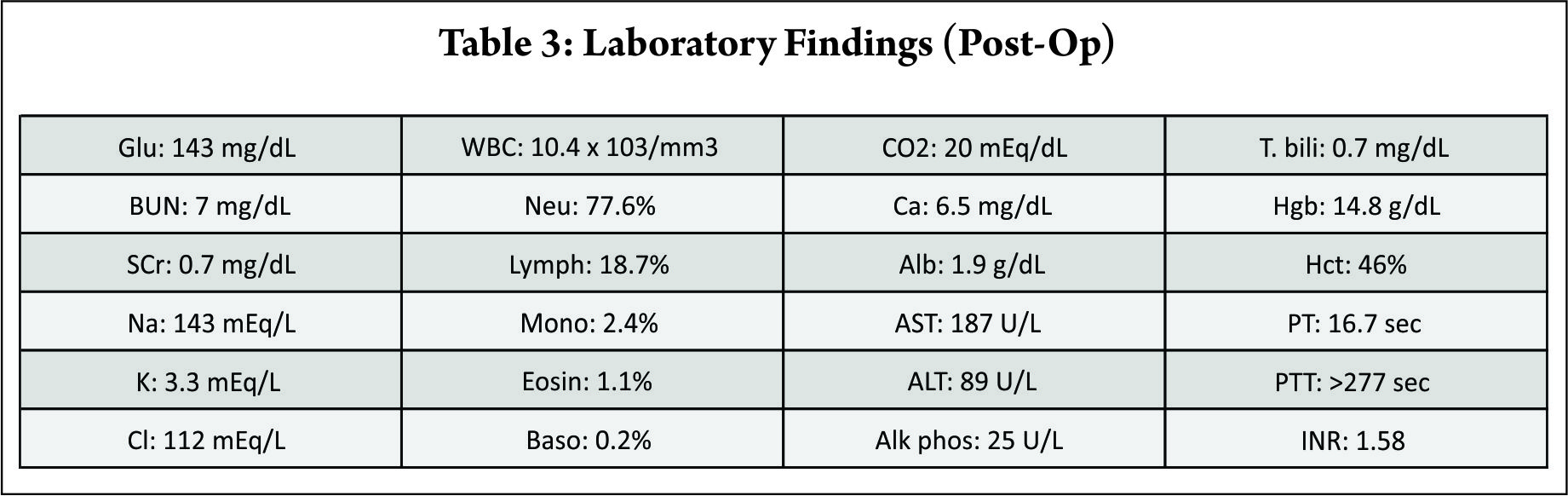
 She denied tobacco, alcohol, and recreational drug use. To her knowledge, she had no previous exposure to Indigotindisulfate. Upon arrival to the operating room, her HR was 80/min, BP 135/75 mm Hg, and a SpO2 of 98%. At 11:30am sedation was initiated using 4 mg of midazolam IV bolus, 100 mcg of fentanyl IV BOLUS, 20 mg of propofol IV BOLUS, and inhaled isoflurane 1.5%-3% with oxygen. After these agents were given her vital signs did not change significantly. She received 2 g cefoxitin IVPB for surgical prophylaxis. A second dose of 100 mcg IV bolus of fentanyl was administered 45 minutes later. The patient’s vital signs remained stable for the next 2 hours (HR = 60-100/min; SBP = 110-150 mm Hg; DBP = 60-80 mm Hg). At 1:15pm the patient received 5 mL of 0.8% Indigotindisulfonate ion via slonjectable solutiw IV bolus over 2 minutes, at which time her BP was 110/60 mm Hg and HR 75/min. Fifteen minutes later the patient became hypotensive, SpO2 97% and the EKG showed sinus bradycardia (BP = 70/50 of mm Hg; HR = 37/min). Despite administering atropine 0.5mg IV BOLUS x2 doses, the patient progressed to pulseless asystole. The team performed the asystole ACLS algorithm, initiating chest compressions, administering epinephrine 1mg IV BOLUS x 4 doses and sodium bicarbonate 50 mEqIV BOLUS x 3 doses with ROSC at 16 minutes. Once the patient was stabilized she received 500mL of 0.9% sodium chloride and 2 units of packed red blood cells. Afterwards the procedure was completed without any other events. During the procedure the patient also received a total of 2.2 L of lactated ringersand suffered an estimated blood loss of 300 mL. The patient was later transferred to the medical intensive care unit in stable condition (BP = 113/75mm Hg, HR=62, O2 sat = 99%, RR =12 breaths/min)(Table 3).
She denied tobacco, alcohol, and recreational drug use. To her knowledge, she had no previous exposure to Indigotindisulfate. Upon arrival to the operating room, her HR was 80/min, BP 135/75 mm Hg, and a SpO2 of 98%. At 11:30am sedation was initiated using 4 mg of midazolam IV bolus, 100 mcg of fentanyl IV BOLUS, 20 mg of propofol IV BOLUS, and inhaled isoflurane 1.5%-3% with oxygen. After these agents were given her vital signs did not change significantly. She received 2 g cefoxitin IVPB for surgical prophylaxis. A second dose of 100 mcg IV bolus of fentanyl was administered 45 minutes later. The patient’s vital signs remained stable for the next 2 hours (HR = 60-100/min; SBP = 110-150 mm Hg; DBP = 60-80 mm Hg). At 1:15pm the patient received 5 mL of 0.8% Indigotindisulfonate ion via slonjectable solutiw IV bolus over 2 minutes, at which time her BP was 110/60 mm Hg and HR 75/min. Fifteen minutes later the patient became hypotensive, SpO2 97% and the EKG showed sinus bradycardia (BP = 70/50 of mm Hg; HR = 37/min). Despite administering atropine 0.5mg IV BOLUS x2 doses, the patient progressed to pulseless asystole. The team performed the asystole ACLS algorithm, initiating chest compressions, administering epinephrine 1mg IV BOLUS x 4 doses and sodium bicarbonate 50 mEqIV BOLUS x 3 doses with ROSC at 16 minutes. Once the patient was stabilized she received 500mL of 0.9% sodium chloride and 2 units of packed red blood cells. Afterwards the procedure was completed without any other events. During the procedure the patient also received a total of 2.2 L of lactated ringersand suffered an estimated blood loss of 300 mL. The patient was later transferred to the medical intensive care unit in stable condition (BP = 113/75mm Hg, HR=62, O2 sat = 99%, RR =12 breaths/min)(Table 3).
On post-op day 1, the patient was successfully extubated and the clinical pharmacist attempted to obtain a medication and allergy history from the patient. However, the patient was not oriented enough to provide any reliable or coherent information. She was found to have a fever and an elevated white blood cell count (WBC) (Table 4). Chest x-ray showed basilar atelectasis and physical exam revealed slightly diminished lung sounds. On post-op day 2, her WBC and temperature both started to decline, and the patient was started on vancomycin 1g IV piggyback once daily, piperacillin/tazobactam 3.375g IV piggyback every 6 hours, and levofloxacin 750mg IV piggyback once daily empirically for suspected healthcare associated pneumonia. On post-op day 3, the patient was transferred to the internal medicine floor, and her WBC and temperature continued to resolve. Her repeat chest X-ray was clear, thus antibiotics were discontinued. A second attempt by the clinical pharmacist to obtain a medication history was made. This time the patient was alert and oriented x 3 and no appreciable motor or neuronal deficiencies were noted or reported by the patient. Patient reaffirmed that she had no significant drug allergies in the past, including over-the-counter and natural remedies. Besides having a shellfish allergy, she denied any food coloring, dyes, or environmental allergies. By post-op day 5 the patient had completely recovered and lungs were clear bilaterally. She was discharged home on metoprolol 50 mg by mouth twice daily, furosemide 20 mg by mouth daily, losartan 25 mg by mouth daily, and albuterol MDI (90 mcg/puff) 1 puff every 4 hours as needed.
Discussion
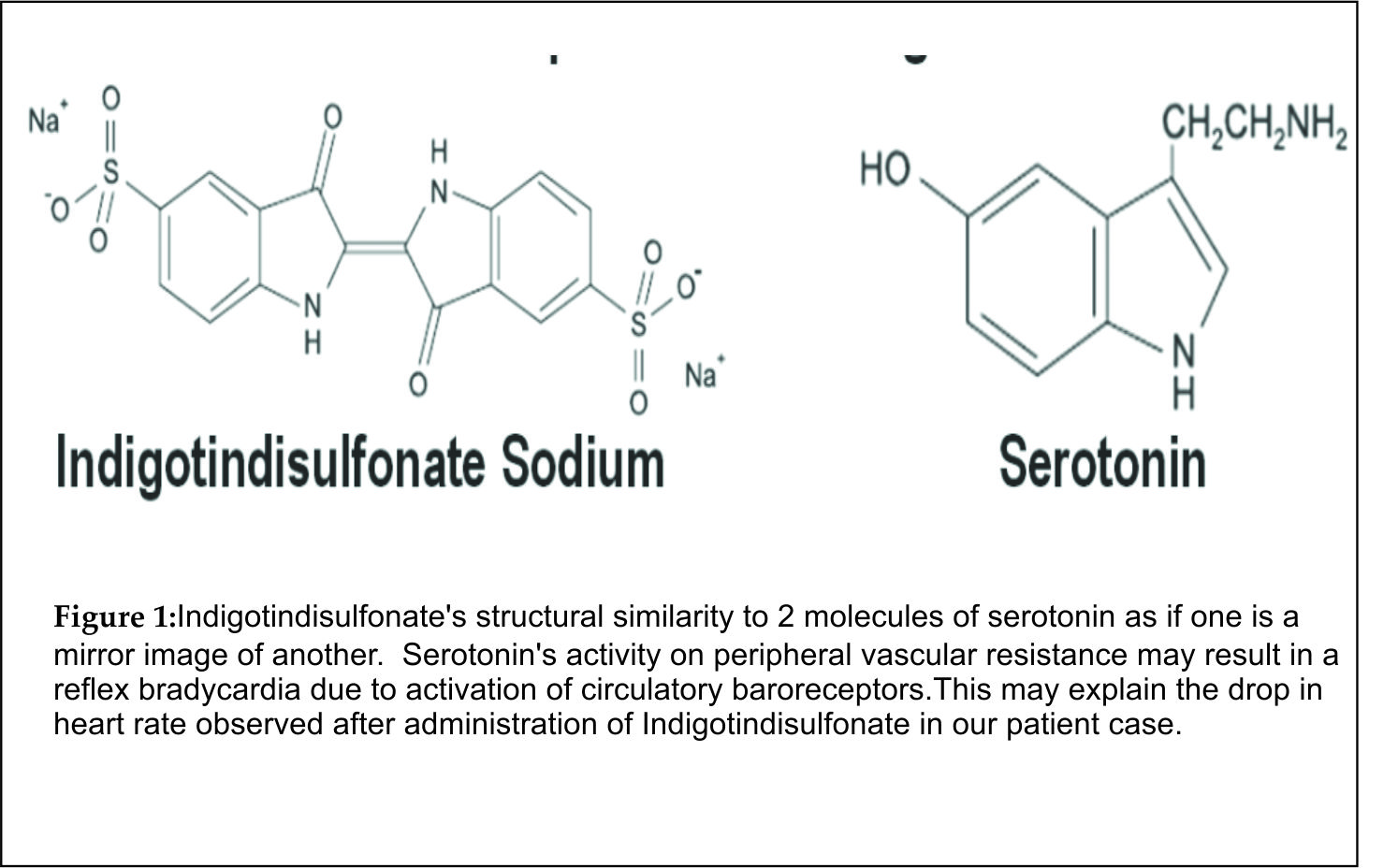
The most likely agent responsible for this patient’s hypotension, severe bradycardia, and eventual cardiac arrest was the Indigotindisulfonate due to the temporal relationship between the onset of adverse drug event and drug administration. To predict the probablility of a true adverse drug raction, the NaranjoNomogram was used[3]. This nomogram was developed in 1981 by Naranjo and colleagues to help standardize assessment of causality for adverse drug reactions. It consists of 10 questions answered yes, no, or “do not know”. Different point values for each question will give the user a score that will be interpreted as doubtful, possible, probable, or definite. The Naranjo Nomogram score was 7, indicating probable Indigotindisulfonate-induced cardiac arrest. Due to the lack of previous exposure or allergies to this drug, this was not a preventable adverse event. Indigotindisulfonate has been widely used as an intravenous injection to localize ureteral orifices during surgery. It is considered safe and efficacious in this setting. However, rare cases of serious adverse events such as cardiac arrest, hypotension, and bradycardia have been reported. Patients often experience a minor transient rise in arterial pressures after intravenous administration of Indigotindisulfonate[4]. Lee and colleagues hypothesized that this is likely due to Indigotindisulfonate’s structural similarity to 2 molecules of serotonin, as if one is a mirror image of another (Figure 1)[5].
Serotonin has been known to increase peripheral vascular resistance, which may result in a reflex bradycardia due to activation of circulatory baroreceptors. This may explain the drop in heart rate observed after administration of Indigotindisulfonate in our patient case. However, this does not explain the sharp drop in her blood pressure. Hypovolemia was ruled out as a possible cause based on the patient’s pre-op vital signs, fluids, and electrolytes, as well as the relatively minor estimated blood loss during the surgery of 300mL.Myocardial infarction or ischemia is also unlikely due to lack of EKG changes during and after the episode. Another theory to this Indigotindisulfonate induced hypotension and bradycardia is possible drug impurities as described in a case series by Kim and colleagues[6]. The only caveat is that the same manufactured lot had been used in other surgeries without any additional events. However, without liquid chromatography and mass spectrometry, this cannot be completely ruled out as a possible cause. Lastly, Shir and Raja reported a series of patients that experienced severe hypotension during radical prostatectomy also citing Indigotindisulfonate as the likely cause due to the temporal relationship[7]. The patients in this case series also received an epidural infusion with local anesthetic, and Fairley theorized that Indigotindisulfonate may induce both stimulant and depressive effects[8]. He proposed that the stimulant effect may have been dulled by the epidural blockade. However, in our case there was no concomitant epidural sympathectomy. Our prevailing hypothesis to the aforementioned adverse effect is an anaphylactoid reaction to the dye causing the release of histamine or other vasodilatory substances. This may explain the hypotension observed in our case, despite the known mild pressor effects of Indigotindisulfonate. Some may argue that the lack of urticaria makes anaphylaxis unlikely, but Brady and colleagues noted urticaria in only 70% of patients treated for anaphylaxis/anaphylactoid reactions[9]. Additionally, coronary vasospasms have been previously associated with anaphylactoid reactions, which may explain the reversible myocardial dysfunction[6]. Although a diagnosis of anaphylaxis requires the evidence more than three organ involvement and detection of specific IgE, or a positive skin test to the alleged agent, these tests were not ordered and therefore we are unable to substantiate for these findings.
Conclusion
In conclusion, Indigotindisulfonate is arelatively safe and widely utilized dye for gynecological procedures. Due to the handful of cases of cardiovascular adverse effects reported with its use, albeit rare, clinicians must still be diligent in monitoring cardiac complications associated with it’s use. Our case report further illustrates that this substance is not completely devoid of adverse effects, and may even lead to a life threatening event. Thus, when cardiac complications arise with its use, health care professionals should be prepared to manage these severe reactions to prevent potentially irreversible ischemic harm to the patient.
Acknowledgements
Cheuk H. (Michael) Liu, Pharm.D., BCPS, BCNSP
Clinical Pharmacist II – HVC/Cardiothoracic ICU Coordinator, PGY-2 Infectious Diseases Pharmacy Residency, Yale-New Haven Hospital.
References
1. Song JE, Kim SK. The use of indigo carmine in ureteral operations. J Urol. 1967; 68:669-70.
2. American Regent. Indigo Carmine (Indigotindisulfonate Sodium) package insert. Shirley, NY; 2011.
3. Naranjo CA et al. A method for estimating the probability of adverse drug reactions. ClinPharmacolTher 1981; 30: 239-245.
4. Wu CC, Johnson A. The vasopressor effect of Indigo Carmine. Henry Ford Hosp Med Journal. 1969;17(2):131-134.
5. Lee WJ , Jang HS. Cardiac arrest from intravenous indigo carmine during laparoscopic surgery -A case report-.Korean J Anesthesiol. 2012 Jan;62(1):87-90.
6. Kim SH , Suk EH, Kil SH, et al. Hypotension in patients administered indigo carmine containing impurities -A case report-. Korean J Anesthesiol. 2011 Nov;61(5):435-8.
7. Shir Y, Raja SN. Indigo carmine-induced severe hypotension in patients undergoing radical prostatectomy. Anesthesiology. 1993 Aug;79(2):378-81.
8. Fairley HB . Hypotension after indigo carmine. Anesthesiology. 1993 Dec;79(6):1454.
9. Brady WJ Jr, Luber S, Carter CT, et al. Multiphasic anaphylaxis: an uncommon event in the emergency department. AcadEmerg Med. 1997(4):193-197.
| How to Cite this Article: Smith AP. A Rare Case Report of Probable Indigotindisulfonate Sodium-Induced Cardiac Arrest. Journal of Anaesthesia and Critical Care Case Reports Jan-Apr 2016; 2(1):8-11. |