Vol 3 | Issue 1 | Jan-Apr 2017 | page: 13-15 | Priyanka Rathi, Arati Ghorpade.
Authors: Priyanka Rathi [1], Arati Ghorpade [2].
[1] Department of anaesthesia, GMC Miraj, pandharpur road, Miraj, 416410.
[2] Department of anaesthesia, RCSMGMC, Kolhapur, 416008.
Address of Correspondence
Dr. Priyanka Rathi
Department of anaesthesia, GMC Miraj,
dist sangli 416410.
Email: priya_rathi76@yahoo.com
Abstract
Introduction: Difficult intubation in a neonate for emergency surgery is a challenge for an anaesthesiologist. We present a case of unanticipated difficult intubation in a newly born female child with tracheoesophageal fistula posted for emergency thoracotomy and repair of fistula. After inhalational induction, laryngoscopy showed clear view of vocal cords but it was impossible to pass an endotracheal tube below the vocal cords. After four attempts of intubation by an experienced anaesthesiologist, narrowing of tracheal lumen was gauged clinically and an emergency tracheostomy was done to secure airway. Post operative CT scan revealed severe congenital subglottic stenosis. This case report describes anaesthesia management of an unexpected failed intubation due to this rare airway defect.
Key words: Congenital subglottic stenosis, tracheostomy, neonate, difficult airway, failed intubation.
Introduction
Incidence of unanticipated difficult airway in children is rare [1]. Most of the airway difficulties are associated with congenital syndromes [2]. Difficult intubation in a neonate posted for emergency surgery is a challenge to anesthesiologist. A physiologically immature neonate with difficult airway and need for emergency surgery is a trio of nightmare for the anesthesiologist. In this article, we present a case of failed intubation in a neonate with tracheoesophageal fistula posted for emergency thoracotomy and repair of fistula and its anaesthesia management.
Case Report
We report a case of an 18 hr old neonate weighing 2.2 kg presented to emergency department with a history of choking, difficulty in breast feeding and increased secretions in mouth.
The baby was delivered by full term vaginal delivery and cried well after birth. The baby was transferred in operation room (OR) wrapped in cotton wool gamzee in radiant warmer receiving oxygen by hood with a patent IV access. Intravenous (IV) fluid 10% dextrose @ 130 cc per 24 hrs was started on admission and antibiotic inj. cefotaxime 10 mg and inj. amikacin 15 mg IV and Inj. vitamin K 1 mg intramuscular (IM) was given. On examination, baby was conscious, warm, pink, afebrile, well hydrated but had a weak cry. Heart rate was 158 beats per min and tachypnoea with respiratory rate of 70 per min. Cardiovascular and Central nervous examination were normal but respiratory system revealed fine crepitations. Routine investigations were normal except blood sugar was 60 milligram (mg) and chest X-ray confirmed tracheoesophageal fistula by presence of coiled ryles tube in blind upper esophageal pouch. The case was classified as American Society of Anaesthesiology (ASA) III physical status and high risk informed consent was taken in view of neonatal emergency and major surgery with perioperative respiratory complications.
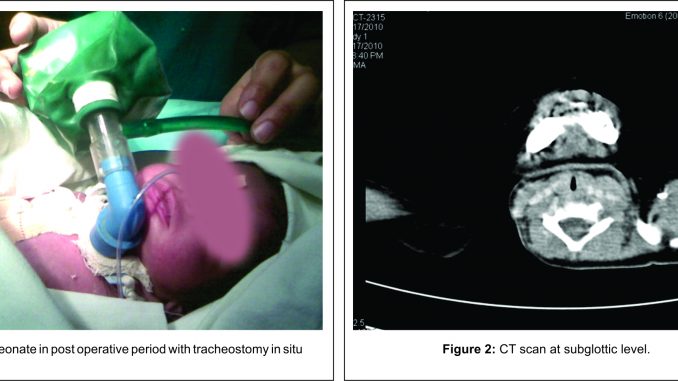
In OR, baby was kept warm and electrocardiogram (ECG), peripheral saturation of oxygen (SPO2) and temperature monitors were applied and basal readings recorded. Premedication was given with inj. atropine 0.05 mg IV. Inhalational induction with oxygen and sevoflurane 3-5% was started. After loss of muscle tone and ensuring deep plane of anaesthesia, laryngocsopy was performed with miller’s blade no 0. Glottic opening was seen, Cormack and Lehane classification was grade 1, so uncuffed portex endotracheal tube no 3 was tried to negotiate through cords but we were unable to pass tube below the cords. So, similar attempt was done with smaller size tubes of no 2.5 and 2.0. After three unsuccessful attempts mask ventilation was done. As mask ventilation was good, we tried fourth attempt with an infant feeding tube of no. 6 but it was very floppy to be negotiated below cords. So, we used a D J stent guide wire as a stylet inside the infant feeding tube. We passed the guide wire below the cords and railroaded the infant feeding tube over it till sufficient length had been reached below cords. It passed easily. Holding the tube at angle of mouth we removed the guide wire and connected the outer end of tube to J R circuit with the help of plastic connecter of no. 2.0 endotracheal tube (ETT). With thumb ventilation, bilateral auscultation confirmed adequate air entry but the infant feeding tube was too soft and flaccid. So a provisional diagnosis of subglottic stenosis or a partial congenital laryngeal web was done. As it was an emergency surgery, emergency tracheostomy was done to secure airway. Tracheostomy was done by ENT surgeon under inhalational anaesthesia with oxygen and sevoflurane 3 % and airway was secured with portex tracheostomy tube no. 3.0. The baby had one episode of convulsion for which inj. phenobarbitone 44 mg IV slowly was given. Surgery was postponed till next day for stabilization of the neonate and received inj. hydrocortisone 4 mg and inj. ranitidine 2 mg and 40 mg paracetamol suppository was kept for overnight analgesia. The next day the case was started as ASA III emergency surgery. Anaesthesia was induced with inj. thiopentone 10 mg IV and maintained on oxygen, sevoflurane 1-2 % and inj. atracurium 1mg IV. Right thoracotomy was done and fistula repaired and intercostal drain (ICD) was kept. Intraoperative all vital monitors were maintained. Fluid was given according to Holiday and Segar formula using 10% dextrose and 40 cc of isolyte P. At the end of surgery intercostal block with 0.25% bupivacaine was given and residual muscle blockade was reversed with inj. neostigmine 0.3 mg and inj. atropine 0.05 mg IV. Paracetamol suppository 20 mg kept for post operative analgesia. Duration of surgery was 45 min and intraoperative blood loss was 5- 10 ml. Post operative the baby was warm pink and moving all limbs and had spontaneous eye opening. The baby was shifted to neonatal intensive care unit (NICU) on a mobile warmer with oxygen at 2 litres (lit) per min through T- piece connected to tracheostomy tube on spontaneous respiration (Fig. 1). On third post operative day, ICD was removed. Tracheostomy tube was removed on fifth postoperative day and after closing of stoma, the baby was discharged on 21st postoperative day. One month later, a computed tomography (CT) scan of neck and thorax was done which confirmed severe subglottic stenosis of 2.8 mm diameter (Fig. 2).
Discussion
Unsuspected subglottic stenosis creating difficult intubating conditions in the operating room is categorized as rare. Congenital subglottic stenosis is an uncommon abnormality that is usually associated with other major or minor abnormalities of the respiratory tract. It is the third most common anomaly of larynx that requires emergency tracheostomy in infants [3, 4]. A subglottic diameter of 4 mm or less in a full-term neonate is considered to be narrowed [5]. Repair of tracheoesophageal fistula is an emergency life saving surgery [6]. Clear plan should be in place to maintain oxygenation and ventilation. At birth, intubation in most full term neonates should be performed with 3.5 mm endotracheal tube. If a smaller than 3.5 mm ETT is required, narrowing of airway may be present. In our case, there was tachypnoea but no stridor to give a clue for possible difficult airway. Awake intubation and inhalational induction are safer options for neonates in emergency surgery. We chose inhalational induction with oxygen and sevoflurane till we obtained deep plane of anaesthesia. Mask ventilation was adequate and oxygen saturation was maintained at 100 %. Laryngoscopy showed clear view of vocal cords, Cormack and Lehane classification grade 1. But we were unable to intubate with smallest possible available 2.0 mm ETT. Inability to advance endotracheal tube beyond cords should have triggered a possibility of narrowing or obstructed tracheal lumen earlier. After three attempts of intubation we decided to use an infant feeding tube no. 6 fr having diameter less than 2.0 mm to assess the patency and lumen of trachea. We were able to pass the feeding tube below the cords by railroading over a double J stent guide wire and connected it to Jackson Rees circuit with plastic connector of no. 2 ETT and confirmed ventilation. Though air entry was adequate, we had a doubt that this infant feeding tube will maintain airway throughout surgery in left lateral position for thoracotomy. The small diameter of infant feeding tube will increase the airway resistance. Similar technique was used by Thampi et al. [7]. Considering the lateral position, risk and duration of surgery we decided to go for a definitive airway i.e. emergency tracheostomy. One episode of convulsion was seen after tracheostomy. As the baby was on spontaneous respiration throughout, we thought the probable cause of convulsion would have been hypoglycemia. Another option of securing airway would have been by use of classic LMA or I gel but we did not have size 1 in our institute. LMA has been recommended as choice of airway in difficult airway in neonates [8]. Tracheostomy, though difficult in neonate was safe option for securing airway in our case. After securing airway, the surgery was uneventful. Post operative CT scan confirmed a congenital subglottic stenosis of 2.8 mm (Fig. 2). Parents were counseled with the diagnosis and a discharge card was given to them with label of a case of difficult intubation due to congenital subglottic stenosis.
Conclusion
Failed intubation in neonate is an important cause of mortality and morbidity during anaesthesia. Presentation of narrow airway in emergency neonatal surgeries is very rare. Primary goal is to achieve safe and rapid stabilization of airway. Prompt diagnosis helps to avoid unnecessary airway manipulations and decrease complications. We could achieve a good outcome with this rare congenital syndrome.
Clinical Relevance
Unanticipated difficult intubation is infrequent in neonates. Laryngeal anomalies are associated with congenital syndromes. Clear view of vocal cords can tempt us for forceful intubation. This will lead to trauma and edema of already compromised airway. Keeping in mind about the possibility of narrow airway due to congenital anomalies, airway should be secured with available equipments and experience of anaesthesiologist. Most important is to provide a discharge card with label as ‘A case of difficult intubation’ and inform the relatives and patient to show it during any medical visit.
References
1. Ramesh S, Jayanthi R, and Archana SR. Paediatric airway management: What is new? Indian J Anaesth. 2012;56(5):448–453.
2. Raj D, Luginbuehl I. Managing the difficult airway in the syndromic child. ContinEducAnaesthCrit Care pain 2015;15:7–13.
3. Daniel SJ. The upper airway: congenital malformations. PaediatrRespir Rev. 2006;7(1) :260-3.
4. Alshammari J, Alanazy S. Optimal Management of a 2-Hour-Old Newborn With Severe Congenital Subglottic Stenosis and Multiple Congenital Heart Diseases. AnnOtolRhinol Laryngol 2015;124:1006-10.
5. Cotton RT. Management of sub glottis stenosis. OtolaryngolClin North Am. 2000; 33(1);111-30.
6. Gupta A. Tracheooesophageal fistula oesophageal atresia &anaesthetic management. Indian J. Anaesth 2002;46(5):353-355.
7. Thampi SM, Salins SR, Jacob DP, Shrestha AS. The Feeding Tube- a Simple Yet Handy Aid to Intubate an Unanticipated Difficult Pediatric Airway. JNMA J Nepal Med Assoc 2015;53(198):141-3.
8. Trevisanuto D, Micaglio M, Ferrarese P, Zanardo V. The laryngeal mask airway: potential applications in neonates. Arch Dis Child Fetal Neonatal Ed 2004;89: F485-F489.
| How to Cite this Article: Rathi P, Ghorpade A. Failed Intubation 1n a Neonate Posted for Emergency Thoracotomy. Journal of Anaesthesia and Critical Care Case Reports Jan-Apr 2017; 3(1):13-15. |

