Vol 5 | Issue 3 | Sep-Dec 2019 | page: 18-20 | Mamta Jain, Vandna Arora, Anil Kumar, Anish K Singh
Authors: Dr. Mamta Jain [1], Dr. Vandna Arora [1], Dr. Anil Kumar [1], Dr. Anish K Singh[1]
Address of Correspondence
1 Department of Anesthesiology and Critical Care, Pandit Bhagwat Dayal Sharma, Post Graduate Institute of Medical Sciences, Rohtak, Haryana, India.
Dr. Vandna Arora,
Pt . BDS PGIMS, Rohtak, Haryana, India.
Email: drvandna4@gmail.com
Abstract
Introduction: Aluminium phosphide is an easily accessible pesticide which is a common cause of suicidal poisoning in developing countries. It is lethal as there is no specific antidote. We came across only one previous case report citing the successful use of intravenous lipid emulsion to treat aluminium phosphide poisoning.
Case report: We present a case of 39 year old male with aluminium phosphide poisoning successfully managed with intravenous intralipid emulsion and magnesium sulphate.
Conclusion: Although the management is primarily supportive, newer management strategies like coconut oil lavage, intralipid emulsion, digoxin use and magnesium sulphate infusion have claimed improved survival.
Keywords: Aluminium phosphide, Intralipid emulsion, Magnesium sulphate
Introduction
Aluminium phosphide is a cheap and effective fumigant pesticide used for both agricultural and non agricultural purposes [1,2]. It is available in the form of tablets, pellets or granules under brand names like celphos, quickphos, alphose etc [1,2]. It causes lethal poisoning by liberating phosphine gas when it comes in contact either with atmospheric moisture or with hydrochloric acid in the stomach. Phosphine causes cellular hypoxia due to its effect on mitochondria, inhibition of cytochrome C oxidase and formation of highly reactive hydroxyl radicals. It particularly affects the cardiac and vascular tissues leading to refractory hypotension, congestive heart failure and eventually multi-organ failure [2,3]. Management is primarily supportive as there is no specific antidote for this poisoning [2]. Newer management strategies like coconut oil lavage, intralipid emulsion, digoxin and magnesium sulphate infusion have claimed improved survival.
Case Report
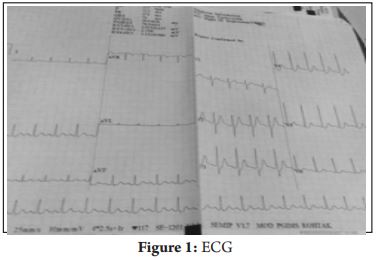
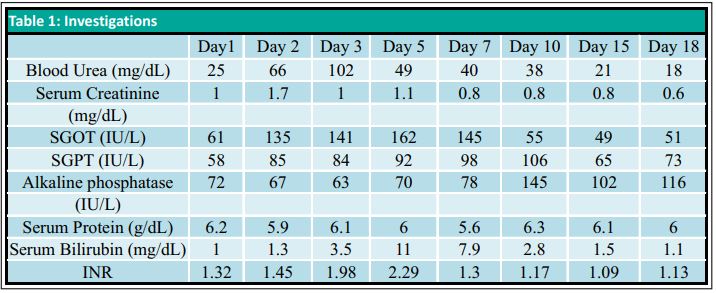
A 39 years old male with alleged history of ingestion of two tablets of celphos presented to the emergency department two hours later with complaints of vomiting and abdominal pain. There was a history of previous suicidal attempt one year back with weedicide spray. On admission, the patient was conscious, oriented with pulse rate of 88/minute, blood pressure (BP) of 80/50 mm Hg, respiratory rate of 25-30/minute and SpO2 of 99% on room air. The patient was resuscitated with intravenous (iv) fluids, gastric lavage was done with potassium permanganate (KMnO4) and was shifted to Intensive Care Unit (ICU) for further management on oxygen by venturimask. Arterial blood gas (ABG) analysis (FiO2 of 0.5) revealed PaO2-164.9, PaCO2-25.9, pH-7.14, HCO3-8.6 and oxygen saturation of 96.4%. Sodium bicarbonate 100 meq was administered slowly (iv) to treat metabolic acidosis and infusion was started with serial monitoring of ABG. Hypotension was managed with iv fluids and dopamine infusion which was gradually tapered and stopped on day 15. The patient was started on intralipid emulsion at 10 ml/hour (iv) within 6 hours of hospital admission with serial monitoring of serum triglyceride levels. Infusion was stopped after 24 hours when serum triglyceride concentration was >500 mg%. Also, magnesium sulfate (MgSO4) 4 g was administered iv over 15-20 minutes followed by 1 g/hour for first 6 hours and then 1 g every 6 hours for 5 days. Signs of magnesium toxicity and serum magnesium levels were closely monitored. MgSO4 dose was reduced to 1 g 12 hourly on day 6 and stopped on day 12.Routine investigations on day 1 revealed hemoglobin of 15.8 g/dl, total leukocyte count (TLC) of 18500/mm3, platelet count of 3.5 lac/mm3, blood urea of 25 mg/dl, serum creatinine of 1.0 mg/ dl, serum sodium of 145 meq/L, and serum potassium of 3.8 meq/L. Liver function tests and coagulation profile were within normal limits. However, over the course of ICU stay, patient developed hepatorenal dysfunction with raised serum bilirubin, liver enzymes, urea and creatinine. Peak derangement was seen on day 5 after which it gradually resolved over time with supportive management.(Table 1)Patient’s ECG showed ST-T changes (Fig. 1) but troponin T was negative and 2D echocardiography revealed normal chamber size and contractility. On day 2, patient’s Troponin T test came positive but repeat echocardiography done on day 3 as well as day 10 was normal. Psychiatric counseling was provided to the patient. Patient’s condition gradually improved after 18 days of management in ICU. Subsequently patient was discharged after two days of observation in the ward.
Discussion
Aluminium phosphide poisoning is a common cause of suicidal poisoning in developing countries due to lack of specific antidote and poor healthcare facilities in rural areas [1,2]. Aluminium phosphide releases toxic phosphine gas on contact with water or hydrochloric acid in stomach which is rapidly absorbed from the gastric mu c o s a [ 2, 3 ]. On reaching th e bloodstream, phosphine causes inhibition of mitochondrial respiratory chain, lipid peroxidation and protein denaturation of cell membrane leading to cell necrosis and release of reactive oxygen intermediates [3-5]. It particularly affects the cardiac and vascular tissues, which manifests as profound hypotension, congestive heart failure, electrocardiographic (ECG) abnorma l iti e s, myocarditis, sub -endocardial infarction or pericarditis [2,3]. Signs and symptoms are nonspecific and may include nausea, repeated vomiting, diarrhea, headache, abdominal pain, breathless ness, tachycardia and hypotension [6,7]. Over time, the patient may develop acute respiratory distress syndrome (ARDS), cardiac arrhythmia, convulsions / coma , hepatorenal dysfunction and eventually multiorgan failure [2,3]. ECG abnormalities include rhythm disturbances, ST-T changes and conduction defects. Metabolic acidosis may be present probably due to the accumulation of lactic acid caused by blockage of oxidative phosphorylation and poor tissue perfusion [6,8]. It may lead to hypoglycemia / hyperglycemia , hypomagnesemia/hypermagnesemia, meth-hemoglobinemia, microangiopathic hemolytic anemia and disseminated intravascular coagulation, although exact pathogenesis is not known [9-11]. Our patient also presented with vomiting, abdominal pain, tachycardia, hypotension with ST-T changes in ECG and developed hepatorenal dysfunction over time. Diagnosis is based on history, garlic breath, and silver nitrate test on gastric aspirate [9,10]. As there is no specific antidote, management is primarily supportive with intensive monitoring of v itals and laboratory parameters. Gastric lavage with saline or sodium bicarbonate or KMnO4 (1:1000) has been recommended by earlier studies. It was suggested that KMnO4 oxidizes phosphine to nontoxic phosphate [3]. However, recently studies have evaluated coconut oil for gastric lavage as it forms a protective layer around damaged gastric mucosa preventing phosphine absorption and also dilutes the gastric acid inhibiting the breakdown of phosphide [12]. We used intralipid emulsion in our case as its successful use has been cited in one previous case report in literature. This is based on “lipid sink” theory which states that toxin with lipid soluble property can be sequestered within the lipid emulsion the rebyreducing its e f f e c t sit e concentration and toxicity [4,13]. The use of soda bicarbonate for moderate to severe acidosis is based on many studies mentioning it as a prognostic indicator of aluminium phosphide poisoning [3]. However, there is literature against the use of sodium bicarbonate also to treat metabolic acidosis in ALP poisoning [14]. Magnesium sulfate has been shown to decrease mortality in these cases in many studies. It acts as a cell membrane stabilizer, reduces the incidence of fatal arrhythmia and combats free radicals [15-19]. Other treatment modalities described are intravenous methyleneblue for methemoglobinemia, N acetylcysteine, digoxin, hyperbaric oxygen, trimetazidine, glutathione, melatonin, vitamin c and boric acid [18-20].
Conclusion
We conclude thatrapid diagnosis, prompt supportive treatment, intensive monitoring and the use of (IV) lipid emulsion and magnesium sulphate can reduce mortality in ALP poisoning. However, large multi centric trialsare needed on these new management strategies.
Clinical relevance
• Aluminium phosphide poisoning is lethal and management is mainly supportive as no specific antidote is available.
• Use of intralipid emulsion and magnesium sulphate can prevent mortality in these cases.
References
1. Vaghefi SS, Emamhadi MA. Aluminium phosphide poisoning: a case report. International Journal of Medical toxicity and Forensic Medicine 2014;4(4):149- 53.
2. Chugh SN. Aluminium phosphide poisoning: Present status and management. J Assoc Physicians India 1992;40:401-5.
3. Gurjar M, Baronia AK, Azim A, Sharma K. Managing aluminium phosphide poisonings. J Emerg Trauma Shock 2011;4(3):378-84.
4. Baruah U, Sahni A, Sachdeva HC. Successful management of aluminium phosphide poisoning using intravenous lipid: Report of two cases. Indian J Crit Care Med
2015;19:735-8.
5. Singh S, Bhalla A, Verma SK, Kaur A, Gill K. Cytochrome-c oxidaseinhibition in 26 aluminium phosphide poisoned patients. Clin. Toxicol. 2006;44(2):155-8.
6. Goel A, Aggarwal P. Pesticide poisoning. Natl Med J India 2007;20:182-91.
7. Sudakin DL. Occupational exposure to aluminium phosphide and phosphine gas? A suspected case report and review of the literature. Hum Exp Toxicol.2005;24:27-33.
8. Gupta S, Ahlawat SK. Aluminium phosphide poisoning-a review. J Toxicol Clin
Toxicol. 1995;33:19-24.
9. Chugh SN, Ram S, Chugh K, Malhotra KC. Spot diagnosis of aluminium phosphide ingestion: An application of a simple test. J Assoc Physicians India 1989;37:219-20.
10. Khosla SN, Nand N, Khosla P. Aluminium phosphide poisoning . J Trop Med IIyg. 1988;91:196-8.
11. Mehrpour O, Jafarzadeh M, Abdollahi M. A systematic review of aluminium phosphide poisoning. Arh IIig Rada Toksikol 2012;63:61-73.
12. Bajwa S, Kaur S, Kaur J, Singh K, Panda A. Management of celphos poisoning with a novel intervention: A ray of hope in the darkest of clouds. Anesth Essays Res 2010;4:20-4.
13. Cave G, Harvey M, Grandins A. Intravenous lipid emulsionas antidote: A summary of published human experience. Emerg Med Australas 2011;23:123-41.
14. Cooper DJ, Walley KR, Wiggs BR, Russel JA. Bicarbonate does not improve hemodynamics in critically ill patients who have lactic acidosis. A prospective, controlled clinical study. Ann Intern Med.1990;112:492-8.
15. Mehrpour O, Farzaneh E, Abdollahi M. Successful treatment of aluminium phosphide poisoning with digoxin: A case report and review of literature. Int J Pharmacol 2011;7:761-4.
16. Chugh SN, Kolley T, Kakkar R, Chugh K, Sharma A. A critical evaluation of antiperoxidant effect of intravenous magnesium in acute aluminium phosphide poisoning. Magnes Res. 1997;10:225-30.
17. Chugh SN, Jaggal KL, Sharma A, Arora B, Malhotra KC. Magnesium levels in acute cardiotoxicity due to aluminium phosphide poisoning. Indian J Med Res. 1991;94:437-9.
18. Karimani A, Mohammadpour AH, Zirak MR, Rezaee R, Megarbane B, Tsatsakis A, Karimi G. Antidotes for aluminium phosphide poisoning-An update. Toxicol Rep. 2018;5:1053-1059.
19. Agrawal VK, Bansal A, Singh RK, Kumawat BL, Mahajn M. Aluminium phosphide poisoning: Possible role of supportive measuresin the absence of specific antidote. Indian J Crit Care Med. 2015;19:109-12.
20. Chugh SN, Arora V, Sharma A, Chugh K. Free radical scavengers and lipid peroxidation in acute aluminium phosphide poisoning. Indian J Med Res. 1996;104:190-3.
| How to Cite this Article:Jain M, Arora V, Kumar A, Singh A. | Intravenous Lipid Emulsion: A Novel Intervention in Management of Celphos Poisoning. | Journal of Anaesthesia and Critical Care Case Reports | Sep – Dec 2019; 5(3): 18-20. |
(Full Text HTML) (Download PDF)
.