Vol 9 | Issue 3 | September-December 2023 | Page: 09-11 | Amruta M Kulkarni, Vijay L Shetty, Gurneet Singh Sawhney
DOI: https://doi.org/10.13107/jaccr.2023.v09.i03.223
Submitted: 10/03/2023; Accepted: 21/06/2023; Published: 10/09/2023
Author: Amruta M Kulkarni [1], Vijay L Shetty [1], Gurneet Singh Sawhney [2]
[1] Department of Anaesthesia, Fortis Hospital, Mulund, Maharastra, India.
[2] Department of Neurosurgery, Fortis Hospital, Mulund, Maharastra, India.
Address of Correspondence
Dr. Amruta M Kulkarni,
Consultant Department of Anaesthesia, Fortis Hospital, Mulund, Maharastra, India.
E-mail: amrutaajgaonkar9@gmail.com
Abstract
Introduction: Language function is complex, involving association between Broca’s motor speech area, Wernicke’s sensory speech area and various interconnected cortical and sub-cortical regions. For lesions in eloquent areas, awake craniotomy with intra-operative neurological monitoring of motor and language function, aids in maximal safe resection of lesion with minimal neurological deficit.
Case presentation: We present a case of 40-year-old patient with left frontal lobe lesion involving motor and speech area who underwent awake craniotomy under scalp block and titrated sedation. Though resection was in safe zone as marked both by neuro-navigation and direct electrical stimulation, patient developed aphasia intra-operatively. The aphasia resolved post-operatively with speech therapy over two weeks. Resection in Supplementary motor area (SMA) in the dominant hemisphere may be the likely cause of aphasia in this patient, resulting in reversible SMA syndrome.
Conclusion: SMA syndrome must be considered as differential diagnosis of deficit during awake craniotomy when resection is in SMA.
Keywords: Aphasia, Supplementary motor area, Awake craniotomy, Eloquent areas
Introduction
Aphasia is a disorder of language function. Main language areas involve Broca’s motor speech area in the inferior frontal gyrus and Wernicke’s sensory speech area in the superior temporal gyrus, with connections between them in form of arcuate fasciculus [1]. Various interconnected streams of cortical and sub-cortical region are involved in speech [1]. For lesions in these eloquent areas, awake craniotomy with intra-operative neurologic monitoring for motor and speech function is preferred, with aim of maximum safe resection [2]. We present a case of 40-year-old patient who underwent awake craniotomy with excision of a large left frontal glioma involving the motor cortex and Broca’s area, with aid of neuronavigation and intra-operative neurologic assessment for motor and speech function to identify the eloquent areas and achieve maximum safe tumor resection.
Case report
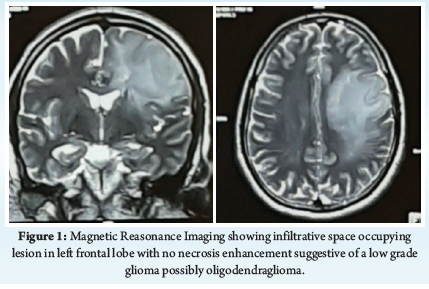
A 40-year-old 63 Kg right handed male patient had chief complains of right sided facial weakness since ten days. Magnetic resonance imaging [MRI] was suggestive of 6.8*4.7*5.8 cm left frontal lobe oligodendroglioma (Figure 1). He was started on anticonvulsant, steroids and, scheduled for an awake craniotomy for excision of left frontal glioma with intra-operative speech and motor function monitoring. After thorough preoperative evaluation and counselling, appropriate consents were obtained. Standard fasting guidelines were followed. Operation theatre was prepared taking into consideration patient condition, and need for awake craniotomy. Standard American society of Anaesthesiology monitoring with depth of anaesthesia monitor-bispectral index [BIS] was initiated. Large bore intra-venous access was secured. Nasal prongs with capnography was used to administer oxygen at 4 litre/min. “Asleep awake asleep” technique was planned. Scalp block was administered with 0.5% bupivacaine to block supra-orbital, supra-trochlear, auriculotemporal, zygomaticotemporal, greater occipital and lesser occipital nerve bilaterally. BIS titrated sedation with infusions of Dexmedetomidine 0.5 mcg/kg/hr and propofol 25-100 mcg/kg/min to maintain BIS in sedation range between 60-80 was administered. Draping was done to accommodate the patient need for neuromonitoring. Dexmedetomidine and propofol infusions were stopped 20 minutes prior to commencement of neuromonitoring and BIS was achieved more than 80 to facilitate neurologic monitoring. Motor and speech area were delineated using neuronavigation and direct electric stimulation with neuro-assessment. Direct electric stimulation was performed with bipolar probe, with biphasic current frequency 60 hertz and amplitude 1-4 mA. Motor function was tested by asking the patient to move right upper and lower limb, while speech was tested by asking to count 1-20 in English, Marathi and answer simple questions. After delineation of motor and speech area, tumor resection was commenced in safe zone with intermittent testing. After about 60% resection of tumor, patient became aphasic but was able to move right upper and lower limbs. No improvement in speech was observed even after waiting for few minutes. Though the surgeon was away from speech area both by neuronavigation and neurologic testing, decision was made to avoid further resection to prevent further deficit. During closure, patient had two focal seizures, first was treated with cold saline irrigation and the second with 30 mg intravenous propofol. Patient was hemodynamically stable throughout the procedure and was shifted to intensive care unit for observation. In immediate post-operative period, patient remained aphasic but hemodynamically stable, seizure free and intact motor function. Post-operative scan showed operative cavity in the left frontal lobe, without any hematoma or infarct. Speech therapy was initiated. Patient was able to speak monosyllables by post-operative day [POD] 2, words by POD 5. Patient was discharged home, and speech therapy was continued. Patient was able to speak complete sentences by POD10 and had no motor deficit or post-operative episodes of seizures.
Discussion
We present a case of awake craniotomy for excision of left frontal glioma involving the motor cortex and Broca’s area. The patient developed aphasia intra-operatively inspite of resection in safe area as demarcated by neuronavigation and neurological assessment with direct cortical stimulation with micro-currents.
Advances in pre-operative functional MRI and intra-operative neuro-navigation aid in accurate localization of eloquent areas, but only awake craniotomy aids in intra-operative assessment of language function [3]. Intra-operative evaluation for speech may done by evaluation of language function, navigation, electrical stimulation of cortex and white matter, and cortico-cortical evoked potentials [3]. Language function is complex with inputs in form of “comprehension” and “cognition” as well as output in form of “articulation” and “utterance”. Hence, localization of language function may be complex and difficult in some cases [3].
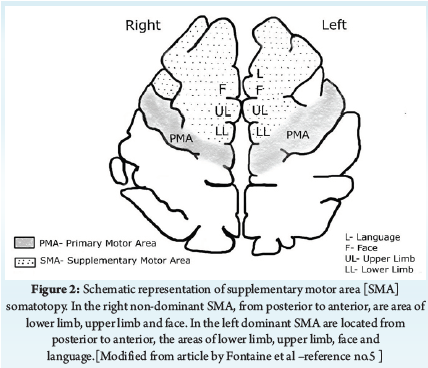
Surgery in the dominant hemisphere may affect the language function. Incidence of immediate post-operative aphasia ranges from 17%-100% [4]. Wilson et al have suggested that aphasia following resection in dominant hemisphere may occur due to the generalized effects of surgery, dysfunction of adjacent or functionally connected regions due to lack of normal inputs from the resected region, transient edema and resection of functional tissue [4]. Our patient developed intra-operative aphasia, though resection was away from speech area, delineated by navigation and intra-operative testing. His post-operative scan showed operative cavity in left frontal lobe, with no hematoma or infarct. Also, aphasia recovered over couple of weeks. In our case, aphasia could have been due to resection in area of supplementary motor area (SMA) in dominant hemisphere. SMA [6] is located in posterior part of the superior frontal gyrus, with cingulated sulcus and precentral sulcus forming inferior and posterior border respectively. The SMA is an eloquent area with rich connection to cortical and sub-cortical structures [5]. Fontaine et al described eleven patients who experienced post-operative neurodeficit after undergoing resection in SMA. One patient had only post-operative aphasia without motor deficit, similar to our case [5]. They suggested in SMA, the representations of the lower limb, the upper limb, the face, and language were located from posterior to anterior in the dominant hemisphere [5] (Figure 2). Resection in SMA in dominant hemisphere, could result in motor and/or speech deficits, similar to SMA syndrome. Deficits correlate with the somatotophy of SMA resection [5]. During awake craniotomy, resection in SMA may cause deficits in areas without positive mapping. Usually, these symptoms are reversible [6]. Nakajima et al described case series of eight patients undergoing awake craniotomy with tumor resection in SMA, of which seven developed post-operative SMA syndrome, which completely reversed in all except one patient [6]. Based on somatotophy of SMA involved in resection, surgeon may be able to predict occurrence of the deficit and reversibility [5]. Patients having SMA region tumors usually undergo awake craniotomy with neuromonitoring, to achieve maximal safe resection, which may result in residual tumor. Kumar et al in their case series of four patients in SMA region tumors, had complete tumor resection under general anaesthesia (GA) with direct electrical stimulation (DES), motor evoked potential monitoring and intra-operative MRI to assess extent of tumor excision. Though all patients developed SMA syndrome post-operatively, it recovered completely. Hence, they suggested that excision of SMA tumors under GA with DES, may increase progression-free survival [7].
Conclusion
Awake craniotomy is preferred technique for resection in eloquent areas of brain to achieve maximum safe resection of lesion. Resection in SMA may occasionally result in motor or language deficit, which is usually reversible, inspite of no positive mapping on testing. These deficits correlate with the somatotophy of SMA resection. We must consider SMA syndrome as differential diagnosis when patients develop deficit in the intra-operative or the post-operative period, when resection is away from eloquent region marked by navigation and neurological testing.
References
[1] Chang EF, Raygor KP, Berger MS. Contemporary model of language organization: an overview for neurosurgeons. J Neurosurg. 2015;122(2):250-261.
[2] Sokhal N, Rath GP, Chaturvedi A, Dash HH, Bithal PK, Chandra PS. Anaesthesia for awake craniotomy: A retrospective study of 54 cases. Indian J Anaesth 2015;59:300-5.
[3] Kanno A, Mikuni N. Evaluation of Language Function under Awake Craniotomy. Neurol Med Chir (Tokyo). 2015;55(5):367-373.
[4] Wilson SM, Lam D, Babiak MC, et al. Transient aphasias after left hemisphere resective surgery. J Neurosurg. 2015;123(3):581-593.
[5] Fontaine D, Capelle L, Duffau H. Somatotopy of the supplementary motor area: evidence from correlation of the extent of surgical resection with the clinical patterns of deficit. Neurosurgery. 2002;50(2):297-305.
[6] Nakajima R, Nakada M, Miyashita K, et al. Intraoperative Motor Symptoms during Brain Tumor Resection in the Supplementary Motor Area (SMA) without Positive Mapping during Awake Surgery. Neurol Med Chir (Tokyo). 2015;55(5):442-450.
[7] Kumar, G.K.; Chigurupalli, C.; Balasubramaniam, A.; Rajesh, B.; Manohar, N. Role of Asleep Surgery for Supplementary Motor Area Tumors. Indian J. Neurosurg. 2022, s-0042-1743266.
| How to Cite this Article: Kulkarni AM, Shetty VL, Sawhney GS. Intra-Operative Supplementary Motor Area Aphosia During Awake Craniotomy a Case Report. Journal of Anaesthesia and Critical Care Case Reports. September-December 2023; 9(3): 09-11. https://doi.org/10.13107/jaccr.2023.v09.i03.223 |
(Full Text HTML) (Download PDF)