Vol 5 | Issue 1 | Jan-April 2019 | page: 9-10 | Meghena Mathew, Nagarajan Ramakrishnan, Ashwin K Mani
Authors: Meghena Mathew [1], Nagarajan Ramakrishnan [1], Ashwin K Mani [1].
[1] Department of critical care medicine
Address of Correspondence
Dr. Ashwin K Mani,
Department of critical care medicine,
Apollo First Med Hospital, Chennai.
Email: ashwin.mani@gmail.com
Abstract
Introduction: We present a rare cause of chronic hypercapnic respiratory failure due to bilateral vocal cord palsy. Identification of chronic bilateral vocal cord palsy as a cause for respiratory failure in an intensive care unit (ICU) can be challenging and the treatment may warrant not only mechanical ventilation but tracheostomy.
Case presentation: A 70-year-old lady presented to us with worsening dyspnea of 10 days duration and stridor of 34 years duration. She had a history of childhood diphtheria. She failed non-invasive ventilation (NIV) for type-2 respiratory failure and required early tracheostomy. Her respiratory failure and clinical condition remarkably improved post tracheostomy. Flexible laryngoscopy showed bilateral vocal cord palsy.
Conclusion: Differentiating the upper airway pathology from obstructive diseases of the lower airway as cause for chronic respiratory failure is essential in a critical care unit, as NIV may not be of any benefit in the former. This case report highlights the importance of considering upper airway disease causing chronic respiratory failure which would require a different clinical management including early intubation with tracheostomy.
Keywords: respiratory failure, vocal cord palsy
Introduction
Bilateral vocal cord palsy is not commonly encountered in Intensive Care Units (ICU) and we could not find any literature. It can cause airway obstruction, leading to dyspnea and respiratory failure. Non-invasive Ventilation (NIV) in this condition may be futile and immediate tracheostomy needs to be considered. We report a patient with childhood diphtheria and bilateral vocal cord palsy of more than 30 years’ duration who presented with breathlessness and acute on chronic type two respiratory failure requiring tracheostomy.
Case Report
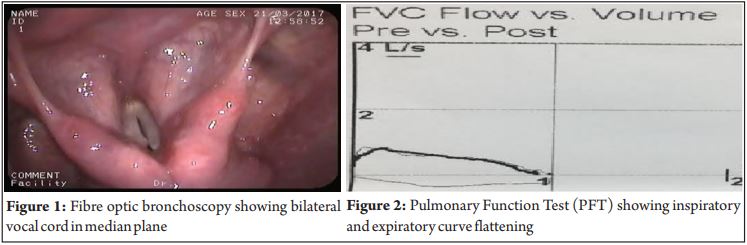
70-year-old lady presented with history of breathlessness of 10 days’ duration and pedal edema for 2 days. She gave history of stridor of 34 years’ duration following childhood diphtheria. Twice surgical correction of vocal cord palsy in the past had failed. On examination, she was in visible respiratory distress. Chest X-ray, Echocardiogram (Echo) and Electrocardiogram (ECG) were normal. Arterial Blood Gas (ABG) revealed acute on chronic type II respiratory failure. Initially she was presumed to have an exacerbation of COPD and was treated with steroids, bronchodilators and Non-Invasive Ventilation(NIV). However, her respiratory acidosis worsened and it was decided to intubate her, followed by tracheostomy. Under sedation and laryngoscopic vision Size 5 ETT was chosen because the patient had a long standing history of stridor and there was ver y narrow vocal cord orifi ce and subsequently tracheostomy done. ABG done two hours’ post procedure showed resolution of type 2 respiratory failure. She was weaned off ventilator support within 24 hours of tracheostomy. Bronchoscopy done after 5 days showed bilateral vocal cord palsy with no movement while attempting to vocalise (Figure 1) . A tri a l of oc c lusion of tracheostomy was not tolerated and it was considered essential to leave the tracheostomy in place. She was discharged with a non-cuffed Shiley tube with a speaking valve with which she was able to eat and speak. Frequent trials of occlusion of tracheostomy attempted. Pulmonary Function Test (PFT) done after one month showed flow volume loop with impairment of inspiratory and expiratory curves (Figure 2). Aft e r two months, she unde r went decannulation successfully
Discussion
We present a patient with chronic respiratory failure following bilateral vocal cord palsy of 34 years’ duration. A review of literature showed that chronic respiratory failure from bilateral vocal cord palsy is rare. Our patient had the longest duration of bilateral vocal cord palsy of 34 years of unknown etiology ever reported. Our review of the literature also showed only one case of post diphtheria neuropathy with vocal cord paralysis with chronic respiratory failure[1]. The diagnosis of bilateral vocal cord paralysis requires a detailed history, clinical examination, pulmonary function test and imaging [2,3]. It is a potentially fatal condition with stridor being one of the presenting history and clinical examination findings. The most common causes for bilateral vocal cord palsy include surger y, tumour or extrinsic compression, intubation, trauma, neurological diseases and radiotherapy[4]. Among tumours, lung malignancies are the commonest[5,6]. Childhood diphtheria is not a common cause of chronic bilateral vocal cord palsy as it recovers spontaneously and does not mandate treatment[1]. Bilateral vocal cord palsy can present with stridor with efficient cough and intact voice. This is due to vibration of vocal cord passively during expiration and speech. Aspiration and dysphagia may not be present with bilateral vocal cord paralysis. If undiagnosed or untreated for a long time, it can result in chronic type two respiratory failure[7] which may be difficult to differentiate from COPD. Our patient had a remote history of surgery for the vocal palsy which was unsuccessful. Our patient presented with history of chronic stridor but acute onset of shortness of breath. Because of this unusual presentation, the recognition of this condition was delayed and she was given trial of NIV. The patients with bilateral loss of vocal cord mobility have a reduced glottic area with increased resistance and any trivial triggers like exercise, inflammation and infection can result in aggravation of stridor, which was very similar to our patient’s presenting complaint. She was otherwise well with history of recurrent worsening of stridor following Upper Respiratory infection (URI). Patients can tolerate this condition for long periods of time until any decompensation occurs. Hence discriminating it from other respiratory failures can be difficult. The most valuable test for diagnosis is flexible fibre optic laryngoscopy which allows direct visualisation of vocal cord and its mobility. Laryngeal ultrasound has also been of value in paediatrics and adult population[8]. The lack of expertise however, limits its use. PFT with inspiratory curve and expiratory curve flattening may be noted in these patients[9]. Diagnosis of chronic bilateral vocal cord palsy as a cause of respiratory failure should be considered in patients who present with chronic stridor. Treatment with NIV may not be beneficial and tracheostomy should be considered. This case highlights the importance of awareness of the role of upper a irway s l e sions in c ausing chroni chypercapnic respiratory failure. Diagnosis of chronic bilateral vocal cord palsy as a cause of respiratory failure should be considered in patients who present with chronic stridor. Treatment with NIV may not be beneficial and tracheostomy should be considered. This case highlights the importance of awareness of the role of upper airways lesions in causing chronic hypercapnic respiratory failure.
Conclusion
Differentiating the upper airway pathology from obstructive diseases of the lower airway as cause for chronic respiratory failure is hence essential in a critical care unit, as NIV may not be of any benefit in the former. This case report highlights the importance of considering upper airway disease causing chronic respiratory failure which would require a different clinical management including early in tubation with tracheostomy.
References
1. Reichler BD1, Scelsa SN, Simpson DM. Hereditary neuropathy and vocal cord paralysis in a man with childhood diphtheria. Muscle Nerve. 2000 Jan;23(1):132-7.
2. Pinto JA, Godoy LB, Marquis VW, Sonego TB. Leal Cde F. Bilateral vocal fold immobility: diagnosis and treatment. Braz J Otorhinolaryngol2011; 77:594–9.
3. Harnisch W, Brosch S, Schmidt M, Hagen R. Breathing and voice quality after surgical treatment for bilateral vocal cord paralysis. Arch Otolaryngol Head Neck Surg2008; 134:278–84.
4. Parnell FW, Brandenburg JH. Vocal cord paralysis. A review of 100 cases. Laryngoscope. 1970;80(7):1036-45.
5. KearsleyJVH.ocal cord paralysis (VCP): an aetiologic review of 100 cases over 20 years. Aust NJM1Zed. 981; 11:663-666.
6. Barondess JA, Pompei P, Schley WS. A study of vocal cord paralysis. Trans Am Clin Clim Assoc. 1985; 97:141- 148
7. Chetty KG, McDonald RL, Berry RB, Mahutte CK. Chronic respiratory failure due to bilateral vocal cord paralysis managed with nocturnal nasal positive pressure ventilation. Chest 1993;103: 1270–1.
8. Amis RJ, Gupta D, Dowdall JR, Srirajakalindini A, Folbe A. Ultrasound assessment of vocal fold paresis: a correlation case series with flexible fiberoptic laryngoscopy and adding the third dimension (3-D) to vocal fold mobility assessment. Middle East J Anesthesiol2012; 21:493–8.
9. Sterner JB, Morris MJ, Sill JM, Hayes JA. Inspiratory flow-volume curve evaluation for detecting upper airway disease. Respir Care 2009; 54:461–6.
| How to Cite this Article: Mathew M, Ramakrishnan N, Mani A K. Chronic respiratory failure from bilateral vocal cord palsy. Journal of Anaesthesia and Critical Care Case Reports Jan-April 2019;5(1):9-10. |
(Full Text HTML) (Download PDF)
.