Vol 8 | Issue 2 | May-August 2022 | Page: 06-11 | Jyoti Shinde, Moosa Awladthani, Sanjay Shinde, Sathiya Murthi, Manthan Shinde
DOI: 10.13107/jaccr.2022.v08i02.199
Author: Jyoti Shinde [1], Moosa Awladthani [1], Sanjay Shinde [2], Sathiya Murthi [3], Manthan Shinde [4]
[1] Department of Anaesthesia & Intensive Care Unit, Armed Forces Hospital, Muscat.
[2] Department of Urology, Armed Forces Hospital, Muscat.
[3] Statistics Specialist, Oman Medical Speciality Board, Muscat.
[4] Maharajgunj Medical Campus, Institute of Medicine, Tribhuvan University, Kathmandu, Nepal.
Address of Correspondence
Dr. Jyoti Shinde
Specialist Anaesthetist, Department of Anaesthesia & Intensive Care Unit, Armed Forces Hospital, Muscat.
E-mail: jyotisanjay@hotmail.com
Abstract
Objectives: To study the sedation challenges in patients with COVID-19-induced acute respiratory distress syndrome.
Methods: A retrospective analysis of sedation challenges in critically ill patients due to COVID-19 pneumonia with acute respiratory distress syndrome at Armed Forces Hospital. Patients requiring continuous infusion or intermittent sedation, or oral/IV sedative therapy, were recorded. The Richmond Agitation and Sedation Scale (RASS) was used as a validated tool to assess sedation. IBM SPSS Version 25 was used for the statistical analysis. Categorical variables were presented with frequency and percentages, as well as continuous mean or SD median or interquartiles.
Results: In total, 100 patients with a mean age of 49.5 ± 14.89 years who recovered, and 67.68 ± 12.75 years who died, were included in the study. To achieve the prescribed sedation level, we observed the need for an intravenous (IV) infusion of two sedatives in 30% of the patients, and three sedatives in 27% of the patients, respectively. Apparently, oral concurrent sedation or analgesia or combinations of these were also prescribed, showing 35% of patients requiring a single drug, whereas 30% required 2-3 drug combinations. The analysis showed that the majority of COVID-19 patients admitted to the ICU required unusually high doses of sedation compared to those available in the literature for non-COVID patients.
Conclusion: The global pandemic outbreak due to SARS CoV-2 continues to affect patients with mild to severe ARDS, requiring ventilation and sedation. High dosages of sedative agents were observed in patients with COVID-19-related ARDS. Several combinations of oral and IV sedatives were used to achieve the required levels of sedation.
Keywords: Respiratory Distress Syndrome, COVID-19, Hypnotics and Sedatives, Pandemics, Analgesia, Intensive Care Units.
Introduction
Corona virus disease 2019 (COVID-19) is a novel disease, which causes severe acute respiratory distress syndrome (ARDS) requiring high quality supportive critical care. During this COVID-19 pandemic, due to development of pneumonia complicated by ARDS, unprecedented number of patients require intensive care unit (ICU) stay [1, 2, 3]. Sedating patients with COVID-19 poses unique challenges in relieving anxiety in non-intubated patients, mechanically ventilated patients and weaning from ventilator.In critically ill patients, the Society of Critical Care Medicine (SCCM) clinical practice guidelines recommend light sedation over deep sedation and nonbenzodiazepines (i.e. propofol or dexmedetomidine) to achieve early extubation and reduce tracheostomy rate and ICU stay [4].It has been reported that critically ill patients with COVID-19 tend to require higher doses of sedation and analgesia. Deeper levels of sedation are required in these patients to prevent patient-ventilator asynchrony, accidental extubations, circuit disconnections, and to facilitate prone ventilation and the use of muscle relaxants in patients with refractory hypoxic respiratory failure [5]. In addition, it also minimizes the nurse’s entry into the patient’s room.However, providing optimal sedation in COVID-19 patients presents unique challenges, not only because of the pathophysiology of the disease, but also because of the unprecedented burden on resources and behavioral changes in healthcare workers to reduce the risk of exposure [6].Moreover, during lung protective ventilation, prolonged sedative infusions are required, leading to gut hypomotility, abdominal distension, and impaired ventilation, increasing the risk of aspiration and also feeding intolerance and malnutrition during prolonged ICU admissions [7]. In addition, prolonged infusions may lead to drug accumulation, tolerance, and tachyphylaxis, along with side effects such as QT interval prolongation and hyperalgesia. Individual features and side effect profiles of sedative drugs, patient factors such as prolonged intubation, and multiple comorbidities must all be considered while managing sedation in COVID-19 ICU patients. However, because a single combination of hypnotic and opioid cannot do this, a sedative strategy including multimodal analgesics and sedatives is required. The analgesia and sedation of all severely ill COVID-19 patients admitted to our intensive care unit were retrospectively analyzed to quantify this issue.
Materials and Methods
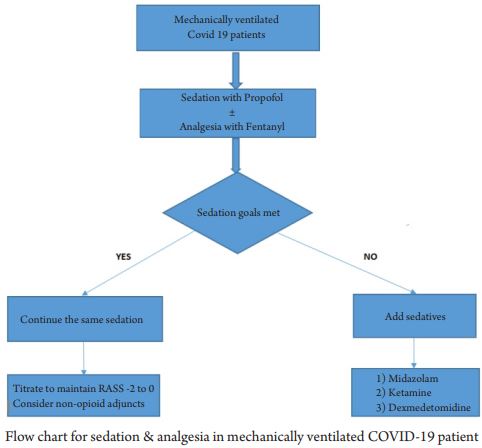
This is a retrospective observational study conducted at the Armed Forces hospital, Muscat. We included all the patients admitted to our intensive care unit (ICU) between April 2020 and December 2020 who were diagnosed with COVID-19 disease with RT-PCR nasal and oral swabs, requiring ICU admission, while patients with negative PCR were excluded from the study. Disease severity was defined by the PAO2/FIO2 ratio and CXR with bilateral infiltrates. Those patients with mild to severe form of disease, admitted to the ICU and requiring oral and IV sedation were evaluated. Sedation was administered as per our ICU standards for critically ill non-COVID ARDS patients. Oral sedation was commonly given to those conscious but anxious patients on NIV with benzodiazepines (Diazepam or Lorazepam) and analgesia with Tramadol in combination with either Haloperidol or Quetipine to avoid delirium. Continuous intravenous (IV) infusion of a strong opioid such as Fentanyl was used as a primary analgesic, along with IV application of sedation. The primary sedatives were propofol or midazolam, and if sedating a patient proved difficult, a combination of the two drugs was administered. In the case of primary use of propofol, conversion to dexmedetomidine or midazolam was initiated to avoid prolonged therapy leading to propofol infusion syndrome.The neuromuscular agent of choice was cisatracurium, which was used only in the case of uncontrollable ventilator asynchrony or during prone position. Types of sedation, including continuous infusion and oral sedation, were noted separately.The Richmond Agitation and Sedation Score (RASS score) was used as a validated and reliable method to assess the patients’ level of sedation in the intensive care unit. In our case, a target RASS of 0 to -1 was aimed as a standard therapy, while in the case of the prone position, a RASS of -3 to -4 was targeted. To determine the side effects associated with the use of other concurrent oral psychotropic drugs such as Haloperidol and Quetiapine, regular ECG was done to assess the QT interval prolongation. Our objectives were to evaluate the type of sedation, requirements, and mode of delivery. The data was analyzed using IBM SPSS Version 25. Categorical variables will be presented with frequency and percentages, as well as continuous mean or SD median or interquartiles.
Results
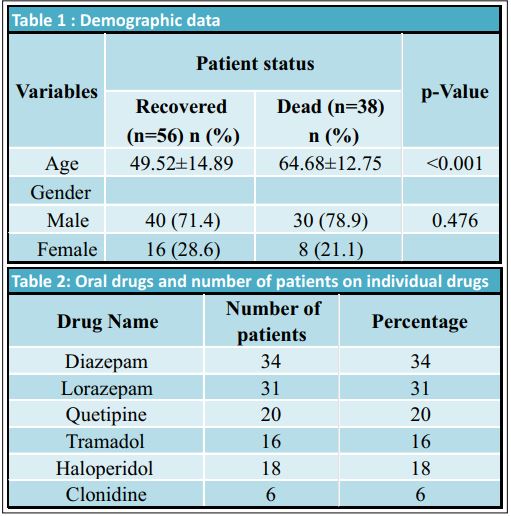
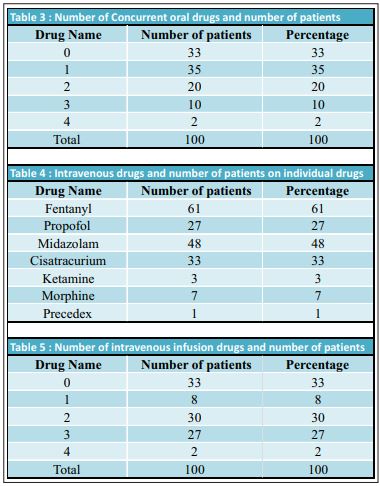
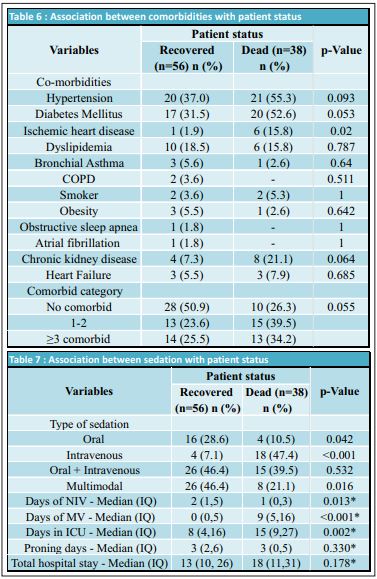
During the study period, we retrospectively assessed 100 patients admitted to the ICU with mild to severe ARDS due to COVID-19 infection. Out of 100 patients, a non-invasive ventilation trial was given to 80, whereas 20 were intubated for mechanical ventilation immediately after ICU admission. Six patients who were declared DNR status (after multidisciplinary evaluation due to the presence of severe disease with multiple comorbidities) were excluded from the study. The demographic data of patients is presented in Table 1.Oral sedation (Table 2) was commonly given to anxious patients with benzodiazepines. Diazepam was prescribed to 34 patients, while 31 received Lorazepam. Analgesia with Tramadol was prescribed to 16 patients, either as a single drug or in combination with psychotropic drugs to avoid delirium, such as Haloperidol in 18 patients, whereas Quetiapine was prescribed to 20 patients. Thirty-three patients did not require any oral sedatives or analgesics. Thirty-five patients were treated with a single agent, while 32 patients required a combination of two to four drugs (Table 3).Mechanically ventilated patients were sedated with Propofol as the first choice and Fentanyl as an anlageiscs. However, when propofol was unavailable due to a shortage, Midazolam was considered as a sedative alternative. Dexmedetomidine infusions as a single drug or in combination with Ketamine continuous infusion were considered in 3 patients. Continuous analgesia was performed with Fentanyl as a primary analgesic in 61% of patients, while Morphine was used as an alternative in seven patients (Table 4). Targeted sedation levels were achieved in 8% of cases by using a single agent, while we found that 30% of patients required a double sedative agent to achieve satisfactory sedation. The majority of these patients received a combination of Fentanyl and Midazolam. Triple sedation combined with an opioid was used to achieve satisfactory sedation in 27% of patients being treated for COVID-19 related ARDS (Table 5). Neuromuscular blocking agent (NMBA) Cisatracurium was prescribed in 33 patients, as the choice of muscle relaxant, for endotracheal intubation, to treat uncontrollable coughing, to give prone position and to improve adequate ventilator synchrony. The graduated sedation depth over the course of the treatment was assessed using the RASS score. We achieved a RASS of 0 to -1 with either single of two drugs, while for those in a prone position, a RASS of -3 to -4 was maintained with NMBA. Associating comorbidities and sedation with patient status is presented in Tables 6 and 7 respectively. All values are Median and Interquartiles with p-Values NIV- Non-invasive Ventilation, IQ- Interquartiles, MV-Mechanical Ventilation, ICU – Intensive Care Unit.
Discussion
COVID-19 is a new disease that causes acute respiratory distress syndrome (ARDS) and necessitates high-quality supportive critical care. Sedating patients with COVID 19 poses unique challenges in relieving anxiety in non-intubated patients, mechanically ventilated patients, and weaning from the ventilator.We evaluated various sedatives and analgesics prescribed either as a single agent or in combination to sedate critically ill COVID-19 ARDS patients.We primarily treated 34 patients with oral benzodiazepines such as Diazepam and 31 patients with Lorazepam, either as a monodrug sedative or as an additional oral sedative to reduce the intravenous infusion. In case of weaning or those on tracheostomy, we also considered using haloperidol or Quetipine to reduce deliriums. Thirty-three patients required no oral sedatives or analgesics. Thirty-five percent were treated with a single medicine, and 32 patients required a combination of two to four drugs.Tramadol, with its multiple mechanisms of action, could be an effective therapeutic option for COVID-19 patients [8].We prescribed Tramadol for 16 patients, either as a single drug or in combination with benzodiazepines or psychotropic drugs such as haloperidol or quetiapine.People with coronavirus disease (COVID-19) may frequently require treatment with psychotropic medications, which will improve the quality of psychiatric care in patients with COVID-19 infection [9]. Another multicenter study suggests that haloperidol use is not associated with any associated risk of intubation or death among adult patients hospitalized for COVID-19 [10]. Short-term use of Haloperidol is generally well tolerated [11], although associated side effects such as QT-interval prolongation and extrapyramidal symptoms can occur [12]. Quetiapine is commonly prescribed for COVID-19 patients with a diagnosis of delirium, but current recommendations are to adjust the dose if patients are on antiretroviral agents [13].In our study, psychotropic drugs were prescribed to avoid delirium with Haloperidol in 18 patients, whereas Quetiapine was prescribed to 20 patients. Well aware of this potential risk of QT-interval prolongation, in our study, daily ECG was done and the QT interval was documented. Only two patients developed QT-interval prolongation, which was picked up early and the psychotropic drugs were immediately discontinued.Several studies reported that for effective sedation with midazolam, a mean dosage of (0.15 mg/kg/hr) was found to be necessary [14, 15]. In our study, midazolam infusion was given for effective sedation at a mean dosage of 5–10 mg/hr.Several studies have found that clonidine at a dose of 1.5 mcg/kg provides adequate sedation when used as a single drug sedative [16, 17]. Clonidine and dexmedetomidine, central a2-agonists, are well known for their co-analgesic and co-sedating properties, which can be beneficial in combination therapies [18, 19].We used oral clonidine for 6 patients with a dose of 1mcg/kg and Dexmedetomidine infusion was considered in only one patient.Propofol infusion is associated with an increased risk of propofol infusion syndrome during long term ventilation and prolonged infusion, hence propofol infusion was discontinued after a few days. Nevertheless, we observed an increased mean dosage of 2-4 mg/kg/min of propofol in comparison to another study reporting a mean dosage of 2.15 mg/kg/min. Also in our study, propofol infusion never exceeded 4 mg/kg/min and was discontinued and replaced with another agent, in addition to monitoring of triglyceride levels.Previously published studies reported the common use of benzodiazepines such as midazolam, propofol, potent opioids, clonidine, or dexmedetomidine, as well as ketamine to achieve the appropriate depth of sedation [20, 21].In our hospital, standards for sedation in critically ill patients appear to be consistent with the published regimens for severe COVID-19 patients in terms of selection of sedation and analgesia. In this study, Fentanyl was given as a primary analgesic rather than a sedative, with Morphine being utilized as an option in seven individuals. Despite the fact that fentanyl has considerable sedative qualities, 59% of patients required a combination of sedatives, highlighting the high level of sedation required during the research period. Endotracheal tube tolerance, invasive catheter insertion, prone positioning, tracheostomy, and chest tube insertion were all made easier with fentanyl. Other than sedatives and opioids, neuromuscular blockade is recommended for patients with ARDS in the first 48 hours after intubation [22, 23].Most of the observations confirm that patients with COVID-19 related ARDS show significant challenges, especially while putting the patient in a prone position [21, 24].In our study, patients on mechanical ventilators in a prone position were prescribed cisatracurium in 33% of patients. Another reason for neuromuscular blocking agent applications was to resolve ventilator asynchronies in spontaneous breathing mode. Few studies have published recommendations for a target RASS of 0 to -1 for the standard sedation therapy in the intensive care unit. Moreover, in the prone position and for ECMO therapy, a RASS of -3 to -4 was targeted in some of the studies [25, 26].Our objective RASS was in line with other studies’ recommendations; we attained a RASS of 0 to -1 with either one or two agents, while those in the prone position maintained a RASS of -3 to -4 with NMBA. When interpreting our results, we faced some limitations. In our study, we included patients admitted to the intensive care unit, either directly from the accident and emergency department or from the COVID ward due to worsening of the disease. Moreover, we did not investigate the history of substance abuse or alcohol, which might have influenced the administration of the sedatives and analgesics. However, our observation of an increased need for sedation in critically ill COVID patients is consistent with the other studies. The main obstacles were the limited knowledge about the pathophysiology of the disease, the lack of clarity about the causes of impaired or increased sedation, and the lack of proper guidelines regarding the sedation strategies in COVID-19 patients. These high requirements were probably related to younger age, good health before infection, intense inflammatory responses in the body, and high respiratory drive [27].We accept that this study was conducted in the early days of the outbreak, having little knowledge about the pathophysiology of the disease. The unusually high requirement for sedation in intensive care unit patients was a surprise to all the clinicians. Over a period of time, it has been demonstrated that the novel coronavirus not only causes lung infection, but it also affects the central nervous system, in addition to many other organs affected. Hence, a hypothesis can be drawn that aggravated sedation could be a consequence of a central nervous system infection. In our retrospective observational study, we found evidence of an unusually high requirement for sedation as well as multiple uses of combination therapies with oral, IV bolus, or continuous infusions, leading to challenges in sedating patients with mild to severe COVID-19 related ARDS. Previously published results from other study groups are in accordance with our observations [28, 29].Future studies are required to address the underlying reasons for the exaggerated high sedation required in patients with COVID-19 related mild to severe ARDS.
Conclusion
The global outbreak continues to affect patients, leading to ARDS and requiring intensive care unit admission for respiratory support and sedation, but optimal sedation strategies are still lacking. Our observations suggest an unusually high dosage requirement of sedatives and analgesics, both for intravenous infusion as well as oral concomitant agents, in patients requiring both non-invasive and invasive mechanical ventilation.
References
1) Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395(10223):497-506.
2) Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513.
3) Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):1-11.
4) Devlin JW, Skrobik Y, Gelinas C, Needham DM, Slooter AJC, Pandharipande PP , et al. Clinical practice guidelines for the prevention and management of pain, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med. 2018;46(9): e825-e873.
5) Kapp CM, Zaeh S, Niedermeyer S, Punjabi NM, Siddharthan T, Damarla M, The use of analgesia and sedation in mechanically ventilated patients with COVID-19 ARDS. Anesth Analg. 2020.
6) Alhazzani W, Evans L, Alshamsi F, Moller MH, Ostermann M, Prescott HC, et al. Surviving Sepsis Campaign Guidelines on the Management of Adults with Coronavirus Disease 2019 (COVID-19) in the ICU: First Update. Critical Care Medicine: March 2021-Volume 49-Issue 3-p e 219-e234.
7) Matos, Renee I. Chung, Kevin K. DoD COVID-19 Practice Management Guide: Clinical Management of COVID-19, Report Date: 2020-06-18.
8) Nahla E El-Ashmawy NE, Abdel-Halim A. Lashin, Kamal M. Okasha, Amal M. Abo Kamer, Tarek M. Mostafa, Mona EL-Aasr, et al. The plausible mechanisms of tramadol for treatment of COVID-19, Med Hypotheses. 2021 Jan;146:110468.Epub 2020 Dec 22.
9) Giovanni Ostuzzi, Davide Papola, Chiara Gastaldon, Georgios Schoretsanitis, Federico Bertolini, Francesco Amaddeo, et al, Safety of psychotropic medications in people with COVID-19: evidence review and practical recommendations. ed. 2020 Jul 15;18(1):215.
10) Nicolas Hoertel, Marina Sanchez-Rico, Raphael Vernet, Anne-Sophie Jannot, Antoine Neuraz, Carlos Blanco, et al. Observational study of haloperidol in hospitalized patients with COVID-19. PLOS ONE, February 19, 2021.
11) Zayed Y, Barbarawi M, Kheiri B, Kheiri B, Banifadel M, Haykal T, et al. Haloperidol for the management of delirium in adult intensive care unit patients: a systematic review and meta-analysis of randomized controlled trials. J Crit Care. 2019;50: 280–286. 10.1016/j.jcrc.2019.01.009
12) Xiong GL, Pinkhasov A, Mangal J, Huang H, Rado J, Jane Gagliardi J, et al. QTc monitoring in adults with medical and psychiatric comorbidities: Expert consensus from the Association of Medicine and Psychiatry. J Psychosom Res. 2020; 110138 10.1016/j.jpsychores.2020.110138.
13) Sampson MR, Cao KY, Gish PL, Hyon K, Mishra P, Tauber W, et al. . Dosing recommendations for quetiapine when coadministered with HIV protease inhibitors. J Clin Pharmacol 2019;59:500–9. 10.1002/jcph.1345.
14) Hanaoka K, Namiki A, Dohi S, Koga Y, Yuge O, Kayanuma Y, et al. A dose-ranging study of midazolam for postoperative sedation of patients: a randomized, double-blind, placebo-controlled trial. Crit Care Med. 2002;30(6):1256–60. Epub 2002/06/20. pmid:12072678.
15) Riker RR, Shehabi Y, Bokesch PM, Ceraso D, Wisemandle W, Koura F, et al. Dexmedetomidine vs midazolam for sedation of critically ill patients: a randomized trial. Jama. 2009;301(5):489–99. pmid:19188334
16) Hayden JC, Bardol M, Doherty DR, Dawkins I, Healy M, Breatnach CV, et al. Optimizing clonidine dosage for sedation in mechanically ventilated children: A pharmacokinetic simulation study. Paediatr Anaesth. 2019;29(10):1002–10. Epub 2019/08/03. pmid:31373752.
17) Srivastava U, Sarkar ME, Kumar A, Gupta A, Agarwal A, Singh TK, et al. Comparison of clonidine and dexmedetomidine for short-term sedation of intensive care unit patients. Indian J Crit Care Med. 2014;18(7):431–6. Epub 2014/08/07. pmid:25097355; PubMed Central PMCID: PMC4118508.
18) Farasatinasab M, Kouchek M, Sistanizad M, Goharani R, Miri M, Solouki M, et al. A Randomized Placebo-controlled Trial of Clonidine Impact on Sedation of Mechanically Ventilated ICU Patients. Iran J Pharm Res. 2015;14(1):167–75. Epub 2015/01/07.
19) Cruickshank M, Henderson L, MacLennan G, Fraser C, Campbell M, Blackwood B, et al. Alpha-2 agonists for sedation of mechanically ventilated adults in intensive care units: a systematic review. Health Technol Assess. 2016;20(25):v–xx, 1–117. Epub 2016/04/02.
20) Wongtangman K, Santer P, Wachtendorf LJ, Azimaraghi O, Baedorf Kassis E, Teja B, et al. Association of Sedation, Coma, and In-Hospital Mortality in Mechanically Ventilated Patients With Coronavirus Disease 2019–Related Acute Respiratory Distress Syndrome: A Retrospective Cohort Study. Critical Care Medicine. 9000;Latest Articles. 00003246-900000000-95286. pmid:33861551
21) Kapp CM, Zaeh S, Niedermeyer S, Punjabi NM, Siddharthan T, Damaria M. The use of analgesia and sedation in mechanically ventilated patients with COVID-19 ARDS. Anesth Analg. 2020.
22) Hraiech S, Forel J-M, Papazian L. The role of neuromuscular blockers in ARDS: benefits and risks. Current Opinion in Critical Care. 2012;18(5):495–502. 00075198-201210000-00013. pmid:22941207
23) Guérin C, Mancebo J. Prone positioning and neuromuscular blocking agents are part of standard care in severe ARDS patients: yes. 2015.
24) Hanidziar D, Bittner EA. Sedation of Mechanically Ventilated COVID-19 Patients: Challenges and Special Considerations. Anesth Analg. 2020: Epub 2020/04/15.
25) Nasraway SA, Jacobi J, Murray MJ, Lumb PD. Sedation, analgesia, and neuromuscular blockade of the critically ill adult: revised clinical practice guidelines for 2002. Critical care medicine. 2002;30(1):117–8.
26) Chanques G, Constantin J-M, Devlin JW, Ely EW, Fraser GL, Gellnas C, et al. Analgesia and sedation in patients with ARDS. Intensive Care Medicine. 2020:1–15. pmid:33170331
27) Martyn JAJ, Mao J, Bittner EA. Opioid tolerance in critical illness. N Engl J Med. 2019;380:365–378.
28) Alhazzani W, Møller MH, Arabi YM, Loeb M, Gong MN, Fan E, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Intensive Care Med. 2020; 46:854–887.
29) Schünemann HJ, Wiercioch W, Brozek J, Etxeandia-Ikobaltzeta I, Mustafa RA, Manja V, et al. GRADE evidence to decision (EtD) frameworks for adoption, adaptation, and de novo development of trustworthy recommendations: GRADE-ADOLOPMENT. J Clin Epidemiol. 2017; 81:101–110.
| How to Cite this Article: Shinde J, Awladthani M, Shinde S, Murthi S, Shinde M | Challenges in Sedating COVID-19 Patients | Journal of Anaesthesia and Critical Care Case Reports | May-August 2022; 8(2): 06-11. |