Vol 4 | Issue 2 | May-Aug 2018 | page: 7-8 | Murlikrishna Kannan, Sushmitha Santhosh.
Authors: Murlikrishna Kannan [1], Sushmitha Santhosh [2].
[1] Anesthesiologist and Founder of Anesthesia Hygiene
[2] Anesthesiologist and Co-Founder of Anesthesia Hygiene
Address of Correspondence
Dr. Murlikrishna Kannan
E-mail: mkannan@anesthesiahygiene.com
Anesthesiologists are well versed in the aseptic nature of surgical procedures. Maintaining sterility of the operating field is in the DNA of every anesthesiologist. In this background, it is intriguing that Infection Control within Anesthesia Work Environment (AWE) garners little to no interest. To most anesthesiologists, Infection Control is changing breathing circuits in between cases, drawing new medications, administering antibiotics, wearing gloves while administering care and performing procedures with universal precautions. There are complex layers of reason for this unique situation. 1. The lack of training in Infection Control. The traditional training of anesthesiologists involves a three to four year residency program that provides exposure to different facets of anesthesiology. Most anesthesiologists are familiar with checklists and protocols for different problems during the peri-operative period. During training, most anesthesiologists get introduced to sterile surgical technique. However there is no formal training or education in Infection Control in the AWE. 2. “What the eye does not see and the mind does not know, doesn’t exist- D.H Lawrence- English Poet.” During the perioperative process,an anesthesiologist frequently encounter problems in a patient- the symptoms and signs of these problems readily manifest. It is a reflex for anesthesiologists to handle these problems. However poor infection control processes do not manifest in any immediate issues for the anesthesiologists. In most cases, patients may be discharged from the hospital and most anesthesiologist may never have a
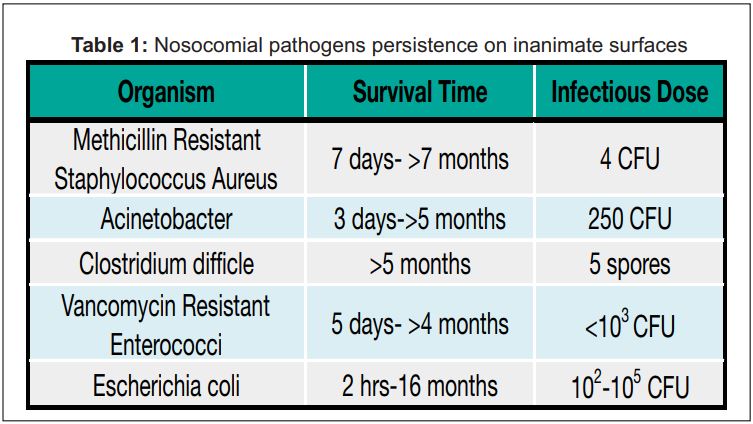
second encounter with the same patient. 3. The lack of resources or tools to improve Infection Control. AWE is contaminated with blood and biological matter from the patient. Clean and dirty equipments are frequently mixed. Clean areas are touched with contaminated gloved hands. Hand Hygiene as per WHO guidelines has been shown to be difficult to adhere during the peri-operative process. 4. The protocol to clean AWE primarily remains wiping the Anesthesia Machine and work surfaces with sanitizing wipes. There is little monitoring, training and validation of cleaning protocols. After a wipe down, there are no checks or validated assessment to ensure AWE is clean. It is important to differentiate between clean versus sterile environment. Sterile environment which is used for surgical environment denotes an absence of microorganism. Clean environment which is generally accepted for AWE means free of dirt, marks or stains to reduce the overall number of organisms[1]. Current Evidences demonstrate that Environmental Hygiene play an important role in development of infections during hospitalization. Patients placed in contaminated rooms or surroundings have a higher chance of acquiring an infection[26]. Several pathogens like Multidrug resistant Acinetobacter, Vancomycin Resistant Enterococci (VRE), pseudomonas are known to exist on surfaces of healthcare facilities or equipment for months [7-14]. Table in an article published by Dancer in Clinical Microbiology Review in 2014 demonstrates this. Do these results translate to anesthesia environment? Loftus et al showed that pathogens on anesthesia machine surfaces directly colonized stopcocks on intravenous sets of patients. In their series of studies they documented incidence of Vancomycin Resistant Enterococci (VRE) transmission via Anesthesia Environment at 32% and two thirds of stopcock contamination had the source from anesthesia environment [15]. In a continuous series of work Loftus et al have demonstrated the transmission of several pathogenic bacteria including MRSA and gram negative bacteria in the Anesthesia Work Environment. Their work has clearly mapped the epidemiology of pathogens in the AWE[16-19]. The evidence points to the need for increased awareness, education and introduction of new practical systems that will prevent contamination and colonization of Anesthesia Work Environment. It is important to recognize the role of the AWE as a reservoir and vector for pathogens potentially leading to infection during the Perioperative period. Hand hygiene is an important methodology to reduce Healthcare Acquired Infections (HAI). However, hand hygiene has been shown to impractical during delivery of Anesthesia and did not reduce contamination of AWE[20]. Anesthesia Hygiene, a company based out of Miami, Florida, USA, designs tools to protect and organize Anesthesia Work Environment to reduce contamination of the AWE. An independent study by Biddle et al quantified the reduction of contamination in the AWE[21]. A larger study will be needed to confirm if this translates into a reduction in perioperative surgical infections.
References
- Rowley S, Clare S, Macqueen S, Molyneux R. ANTT v2: an updated practice framework for aseptic technique. Br J Nursing. 2010;19:S5–S11
- Dancer SJ. 2008. Importance of the environment in Methicillin-resistant Staphylococcus aureus acquisition: the case for hospital cleaning. Lancet Infect. Dis. 8:101–113. 10.1016/S1473-3099(07)70241-4
- Martinez JA, Ruthazer R, Hansjosten K, Barefoot L, Snydman DR. Role of environmental contamination as a risk factor for acquisition of vancomycin-resistant enterococci in patients treated in a medical intensive care unit. Archives Int. Med. 2003 163:1905–1912. 10.1001/archinte.163.16.1905
- Tankovic J, Legrand P, de Gatines G, Chemineau V, Brun-Buisson C, Duval J.Characterization of a hospital outbreak of imipenem-resistant Acinetobacter baumannii by phenotypic and genotypic typing methods. J. Clin. Microbiol. 1994 32:2677–268
- Green J, Wright PA, Gallimore CI, Mitchell O, Morgan-Capner P, Brown DWG.The role of environmental contamination with small round structured viruses in a hospital outbreak investigated by reverse-transcriptase polymerase chain reaction assay. J. Hosp. Infect. 1998 39:39–45. 10.1016/S0195-6701(98)90241-9
- Kaatz GW, Gitlin SD, Schaberg DR, Wilson KH, Kauffman CA, Seo SM, Fekety R. Acquisition of Clostridium difficile from the hospital environment. Am. J. Epidemiol. 1998. 127:1289–1294
- Kramer A, Schwebke I, Kampf G.How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006 6:130. 10.1186/1471-2334-6-130
- Wagenvoort JHT, Sluijsmans W, Penders RJR. Better environmental survival of outbreak vs.sporadic MRSA isolates. J. Hosp. Infect. 2000 45:231–234. 10.1053/jhin.2000.0757
- 9. Chiang SR, Chuang YC, Tang HJ, Chen CC, Chen CH, Lee NY, Chou CH, Ko WC. Intratracheal colistin sulfate for BALB/c mice with early pneumonia caused by carbapenem-resistant Acinetobacter baumannii. Crit. Care Med. 2009 37:2590–2595. 10.1097/CCM.0b013e3181a0f8e1
- Lawley TD, Clare S, Deakin LJ, Goulding D, Yen JL, Raisen C, Brandt C, Lovell J, Cooke F, Clark TG, Dougan G.Use of purified Clostridium difficile spores to facilitate evaluation of health care disinfection regimens. Appl. Environ. Microbiol. 2010. 76:6895–6900. 10.1128/AEM.00718-10
- Kjerulf A, Espersen F, Gutschik E, Majcherczyk PA, Hougen HP, Rygaard J, Høiby N. Serological diagnosis of experimental Enterococcus faecalis endocarditis. APMIS 1998. 106:997–1008. 10.1111/j.16990463.1998.tb00252.
- Teunis PF, Moe CL, Liu P, Miller SE, Lindesmith L, Baric RS, Le Pendu J, Calderon RL. Norwalk virus: how infectious is it? J. Med. Virol.2008. 80:1468–1476. 10.1002/jmv.21237
- Eaton KA, Friedman DI, Francis GJ, Tyler JS, Young VB, Haeger J, Abu-Ali G, Whittam TS. 2008. Pathogenesis of renal disease due to enterohemorrhagic Escherichia coli in germ-free mice. Infect. Immun.76:3054–3063. 10.1128/IAI.01626-07
- Wandall DA, Arpi M, Wandall JH. 1997. A rat model of non-lethal bacterial infection. APMIS 105:187–191. 10.1111/j.1699-0463.1997.tb00557.
- Loftus RW, Koff MD, Burchman CC, Schwartzman JD, Thorum V, Read ME, Wood TA, Beach ML. Anesthesiology 2008. 109(3): 399-407
- Loftus RW, Koff MD, Brown JR, Patel HM, Jensen JT, Reddy S, Ruoff K, Heard SO, Yeager MP, Dodds TM. The Epidemiology of Staphylococcus Aureus Transmission in the Anesthesia Work Area. Anesthesia & Analgesia 2015. Vol 120, No. 4, 807-818
- Loftus RW, Koff MD, Brown JR, Patel HM, Jensen JT, Reddy S, Ruoff K, Heard SO, Yeager MP, Dodds TM. Transmission Dynamics of Gram Negative Bacterial Pathogens in the Anesthesia Work Area. Anesthesia & Analgesia 2015. Vol 120, No. 4, 819-826
- Loftus RW, Koff MD, Brown JR, Patel HM, Jensen JT, Reddy S, Ruoff K, Heard SO, Yeager MP, Dodds TM. TheDynamics of Enteroccocus Transmission from the Bacterial Reservoirs Commonly encountered by Anesthesia Providers. Anesthesia & Analgesia 2015. Vol 120, No. 4, 827836
- Loftus RW, Koff MD, Birnbach DJ. The dynamics and Implication of Bacterial Transmission events arising from the Anesthesia Work Area. Anesthesia & Analgesia 2015. Vol 120, No. 4, 853-860.
- Rowlands J, Yeager MP, Beach M, Patel HM, Huysman BC, Loftus RW. Video Observation to map hand contact and bacterial transmission in operating rooms. Am Journal of Infection Control 2014. 42: 7; 698-701
- Biddle CJ, Gay BE, Prasanna P, Hill EM, Davis TC, Verhulst B. Canadian Journal of Infectious Disease and Medical Microbiology 2018. Article ID 1905360
| How to Cite this Article: Kannan M, Santhosh S. Anesthesia and Infection Control. Journal of Anaesthesia and Critical Care Case Reports May-Aug 2018; 4(2):7-8. |
(Abstract) (Full Text HTML) (Download PDF)
.