Vol 3 | Issue 2 | May-Aug 2017 | page: 30-33 | Christopher J. Mallard, Habib Srour
Authors: Christopher J. Mallard [1], Habib Srour [1]
[1] Department of Anesthesiology, University of Kentucky Medical Center, Lexington, KY 40536-0293, USA..
Address of Correspondence
Christopher J. Mallard,
Department of Anesthesiology, University of Kentucky Medical Center
Lexington, KY 40536-0293, USA. Phone: +(859) 218-4838.
Fax: +(859) 323-1080.
E-mail: cjmallard@uky.edu
Abstract
Introduction: Post-operative stridor is a common respiratory complication seen in the post-anesthesia care unit. Stridor can be a symptom of an anatomic, obstructive of metabolic cause. However, when conventional treatments fail further investigation is warranted. Our patient was found to have a non-anion gap metabolic acidosis (NAGMA) secondary to renal tubular acidosis (RTA), which once corrected resulted in immediate resolution of her stridor. The literature has presented case reports of hypokalemia-induced stridor; however, to the best of our knowledge, there have been no reports of acidosis secondary to RTA as a cause for stridor.
Case Report: A 65-year-old caucasian female female with a history of endometrial cancer, chronic kidney disease, and a known mediastinal mass who is seen following repair of a pathologic femur fracture. After failing conventional treatments for upper airway obstruction, she was found to have a NAGMA consistent with RTA. The patient compensated for the acidosis by hyperventilating but due to the tracheal stenosis from her mediastinal mass developed a flow-dependent stridor. Once the acidosis was treated with sodium bicarbonate the patient had immediate resolution of the stridor.
Conclusion: This case study can be used as a learning tool as a broad differential diagnosis was necessary in determining the cause of a relevantly common post-operative complication.
Keywords: Stridor, metabolic acidosis, renal tubular acidosis, mediastinal neoplasms.
Introduction
Stridor can be a symptom of different pathologies including anatomic, obstructive, or metabolic. We present a case where the patient developed stridor secondary to the combination of renal tubular acidosis (RTA) and an obstructive hilar mass. It was only after she did not respond to typical treatments for stridor that the cause of her acidosis was determined and treated with bicarbonate. Following bicarbonate administration, the patient had an immediate resolution of the stridor.
Case Report
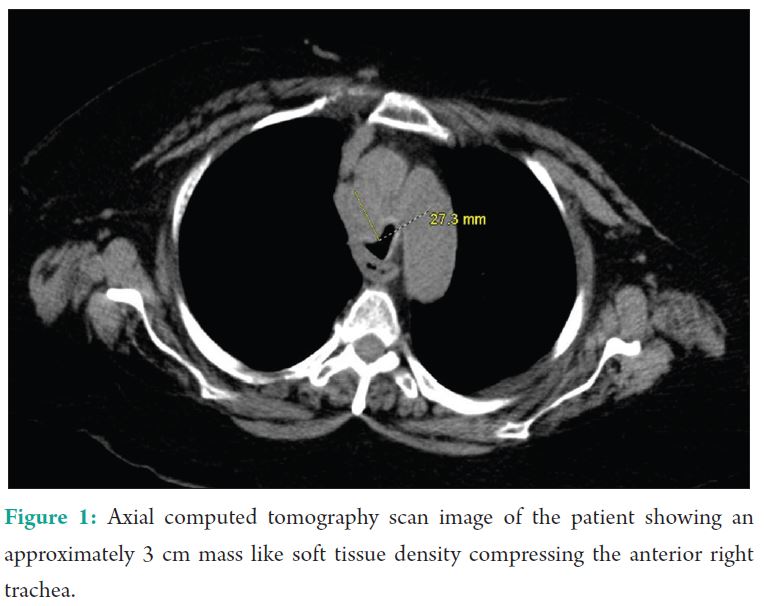
This patient is a 65-year-old female with history of metastatic Stage IIIB endometrial cancer, tracheal stenosis secondary to a paratracheal mass/lymph node, right hilar mass (Fig. 1), chronic kidney disease (CKD) Stage 3B, asthma, and multiple malignancy-associated deep vein thromboses who presents with right leg pain proximal to the knee after a fall.
Before presentation, the patient had been walking normally and had suddenly heard a “pop” in the right leg with a subsequent fall to the ground. She was admitted to the orthopedic surgery service for a pathologic femur fracture and was found to have a pre-renal acute kidney injury. 3 days after admission, the patient was taken to the operating room (OR) for a distal femur repair.
She was evaluated preoperatively by anesthesiology and was classified as an American Society of Anesthesiologists physical class four and planned for general endotracheal anesthesia. Neuraxial anesthesia could have been utilized as the patient’s international normalized ratio was 1.2 on the morning of surgery and prophylactic dose enoxaparin had been held over 12 h; however, given the patient’s body habitus, complexity of surgery, concerns over the ability to lie flat for an extended period of time, and most importantly – patient refusal, the anesthesia team elected to forgo regional anesthesia. She was given midazolam and taken to the OR. She received albuterol for wheezing and underwent induction of general anesthesia with intravenous (IV) fentanyl, lidocaine, propofol, and rocuronium 30 mg. Intubation required two attempts due to the practitioner’s inability to pass a 7.5 endotracheal tube (ETT) but was successful with a 6.5 ETT. Following intubation, a radial arterial line and central venous cannula were placed. Maintenance of anesthesia was with sevoflurane and pain was controlled with fentanyl and hydromorphone boluses. She did require the initiation of a phenylephrine infusion 30 min into the case to maintain mean arterial pressures in the 70’s. Rocuronium was dosed with another 10 mg approximately 30 min after the initial bolus with induction. Train-of-four ratios was not recorded during the case; however, approximately 1 h following the second dose of rocuronium, the patient was ventilated on pressure support of 10 and positive end expiratory pressure of 5. She received reversal of neuromuscular blockade with 5 mg of neostigmine and 0.8 mg of glycopyrrolate 15 min after starting pressure support. The case took longer than expected after reversal had been given since the femur required a larger than normal resection. The total knee replacement was performed with a distal femoral hinged prosthesis which is also a more laborious surgical procedure than standard total knee replacement. However, given the ease of intubation and adequate tidal volume on minimal pressure support the anesthesia teams made the decision to extubate in the OR and forgo post-operative ventilation. 70 min following reversal of neuromuscular blockade the patient received two puffs of albuterol and extubated to 10 L of oxygen through face tent in the OR. She was transported from the OR awake, following commands, and hemodynamically stable off phenylephrine. Initial vital signs in the post-anesthesia care unit (PACU) were blood pressure 113/70, heart rate 99, respiratory rate 15, oxygen saturation 99%, and temperature of 35.7°C. Approximately 20 min following extubation, she developed audible stridor with bilateral lung wheezing on exam with no tachypnea, desaturation, or complaints of respiratory distress. She received 8 mg of dexamethasone 5 min after the stridor developed. 7 min later, she received nebulized albuterol and ipratropium. The stridor continued and 6 min after the albuterol/ipratropium, she received a dose of nebulized racemic epinephrine. The stridor did not respond to any of these interventions. Train-of-four was not performed in the PACU given the patient’s clearly adequate grip strength. Fiberoptic bronchoscopy was discussed but not utilized during the patient’s PACU stay as she had stable oxygen saturation and PaO2 on arterial blood gas (ABG) and the team did not feel the risk of sedation and bronchoscopy outweighed the potential benefits. Resolution of the signs and symptoms with medical management in the setting of a known obstruction was sufficient. During this time, her oxygen saturation remained over 98% on 10 L/min face tent with respiratory rates of 14-18 breaths/min. She continued to have audible stridor with improved wheezing and 2 h after the stridor began, she was started on high flow nasal cannula at 20 L/min FiO2 of 30%. She continued on high flow nasal cannula with respiratory rate ranging 13-21 breath/min and oxygenation above 96%. Upon review of her laboratory work, ABG in the OR showed a pH – 7.33, PaCO2 – 36 mmHg, PaO2 – 289 mmHg, HCO3- 19 mmol/L, FiO2 – 94%, and in PACU 3 h later pH – 7.23, PaCO2 – 40 mmHg, PaO2- 81 mmHg, HCO3- 17 mmol/L, and FiO2 – 10 L face tent. Her most recent basic metabolic panel had results of Na – 136 mmol/L, chloride – 103 mmol/L, HCO3 – 19 mmol/L, and anion gap 14. These laboratory values were consistent with a non-anion gap metabolic acidosis (NAGMA) prompting urine electrolytes to be obtained which were consistent with a RTA (urine Na – 83 mmol/L, urine K – 34 mmol/L, urine Cl – 75 mmol/L, and urine anion gap positive). The patient was given 50 mEq of sodium bicarbonate approximately 4 h after the stridor had begun with immediate resolution. The patient was admitted to the intensive care unit overnight for monitoring without any further episodes of stridor and was placed on a scheduled oral dose of sodium bicarbonate (650 mg 4 times daily). Bicarbonate was trended with basic metabolic panels postinitiation of replacement therapy, given that it is a directly measured value on this panel and her pH had demonstrated normalization with IV therapy in the PACU. Her initial postoperative serum bicarbonate was 15 mmol/L which was obtained 6 h after the PACU ABG. 2 h later repeat bicarbonate was 16 mmol/L. 10 h later bicarbonate increased to 18 mmol/L and returned to normal levels at 23 mmol/L on the morning of post-operative day 2. She was discharged on post-operative day 5 to an acute care rehabilitation center after resolution of her RTA and no further need for bicarbonate supplementation.
Discussion
The Stridor is caused by turbulent airflow through a partially obstructed airway leading to high-pitched sounds which can be extremely variable. The obstruction can occur at multiple levels in the supraglottis, glottis, subglottis, or trachea. Figure 1: Axial computed tomography scan image of the patient showing an approximately 3 cm mass like soft tissue density compressing the anterior right trachea. Stridor occasionally results from swelling secondary to epithelial damage related to the ETT. This results in a decreased ccross-sectional diameter and increased pressure gradients during breathing. The smaller cross-sectional diameter of the airways increases the turbulence of the airflow leading to increased work of breathing, respiratory rate, and metabolic demands. Treatments for stridor include humidified oxygen, helium/oxygen mixtures, sitting upright, IV dexamethasone, and/or nebulized racemic epinephrine [1]. Airway edema is a frequent complication of intubation typically presenting within 30 min after extubation. The edema is secondary to the inflammatory response from the ETT causing pressure and ischemia resulting in mucosal injury. Risk factors for laryngeal injury, edema, and post-extubation stridor have included female gender – possibly due to less resistant mucus membranes – and larger tube to trachea ratio. However, there is still controversy about the length of time spent intubated and post-extubation edema and stridor [2]. A common cause of stridor in the PACU is laryngospasm. Laryngospasm occurs when there is reflex constriction of the intrinsic laryngeal muscles resulting in glottic closure and decreased open area through which air may enter the trachea. Management of laryngospasm includes positive pressure, succinylcholine, small dose propofol, magnesium, nitroglycerin, and topical or aerosolized lignocaine [3]. Residual neuromuscular blockade is a commonly observed complication in the PACU with reports of up to 64% of patients having inadequate neuromuscular recovery. Train-of-four ratio of <0.7-0.9 is associated with upper airway obstruction, inadequate ventilation, reduced pharyngeal muscle coordination, as well as an increased risk of aspiration. In addition, multiple investigations have noted an associated between hypoxemia (SpO2<90%) and residual neuromuscular blockade [4]. Paradoxical vocal cord motion (PVM) is a known cause of non-organic upper airway obstruction leading to stridor that is diagnosed with direct visualization of vocal cords. In patients with PVM, there is adduction of the vocal cords during inspiration and abduction during expiration. Patients are typically treated with sedating agents such as benzodiazepines since there is usually a psychological component [5]. Other less common causes can be metabolic in origin such as hypokalemia, hypophosphatemia, or hypocalcemia. Botha et al. reported a case where stridor and respiratory distress occurred secondary to hypokalemia and hypophosphatemia after profound diarrhea. They hypothesized that muscle weakness caused by hypokalemia and hypophosphatemia were the metabolic causes for the patient’s respiratory distress and stridor [6]. Moralee and Reilly report a case where hypokalemia led to bilateral vocal cord paralysis which is typically a complication after thyroidectomy. The authors, however, were unable to find a specific link in the literature to associate hypokalemia and vocal cord paralysis [7].
RTA is a group of disorders where – even with preserved glomerular filtration rate – patients develop a NAGMA. There are three different types broken down into either the area of the affect or related to aldosterone. Distal RTA (Type 1) is caused by either a tubular membrane defect or defect in the hydrogen pump itself preventing hydrogen from getting into the urine to produce ammonium. An indirect way to estimate the urinary ammonium
excretion is to look at the urine anion gap (Na+ + K+ − Cl−); negative values reflect high urinary ammonium which would be expected in patients that have an acidosis and normal tubular function. The urine anion gap is positive in distal RTA because of the inability of the tubules to secrete hydrogen ion and make ammonium. Hypokalemia caused by distal RTA is typically multifactorial: Volume contraction, increased activity of the renin-angiotensin aldosterone system, and substitution of potassium for hydrogen ion to exchange for distal tubular sodium. Patients typically present with musculoskeletal complaints secondary to the hypokalemia. Another distinct presentation of patients with Type I RTA is stone production secondary to the hypercalciuria from bone buffering. Proximal or Type 2 RTA is the least common form and is characterized by dysfunction of the proximal tubular transporter leading to reduced absorption of bicarbonate and significant bicarbonaturia. However, with proximal RTA once a threshold bicarbonate level in the plasma has been reached the proximal tubule can reclaim some of filtered bicarbonate. Because the distal acidification system remains intact, these nephrons are able to excrete more acid to the urine. Thus, these patients typically do not accumulate as much as acid as the Type I or Type 4 varieties. Type 4 RTA typically has associated hyperkalemia secondary to decreased aldosterone secretion or aldosterone resistance. Aldosterone typically causes sodium independent hydrogen secretion into the urine. However, when aldosterone is not present there is a decreased electrical stimulus for hydrogen secretion [7, 8, 9]. Medications can also be a cause of RTA including alkylating agents, aminoglycosides, anti-epileptics, anti-protozoal, dicarboxylic acids, iron chelators, nucleoside analog reverse transcriptase inhibitors, nucleotide analog reverse transcriptase inhibitors, platinum containing compounds, salicylates, and tetracycline antibiotics. This patient had been treated with platinum containing compounds in the past (carboplatin) and at the time of admission was on an alkylating agent (cyclophosphamide). Both of these drugs classes are taken into the proximal tubular cells leading to damage. Recovery of proximal tubular cell function can take months and may not always be complete. There have been reports of long term follow-up patients on ifosfamide where tubular dysfunction remains 10 years posttreatment [10, 11].
Conclusion
In general, for anesthesiologists: Hypovolemia and aldosterone dysregulation caused by positive pressure ventilation can contribute to mild forms of RTA. In this patient, the cause was likely combination of cancer treatment and CKD. She compensated for this acidosis through hyperventilation. However, given her mediastinal mass, the increased respiratory rate needed to compensate for the RTA led to the development of flow dependent stridor. The patient’s stridor resolved immediately with bicarbonate administration because she no longer felt compelled to hyperventilate to compensate for the NAGMA.
References
1. Barash PG, Michael KS, Stock MC, Ortega R, Stock C, Ortega R. Clinical Anesthesia. 7th ed. Philadelphia, PA:
Lippincott Williams & Wilkins; 2013.
2. Wittekamp BH, van Mook WN, Tjan DH, Zwaveling JH, Bergmans DC. Clinical review: Post-extubation laryngeal
edema and extubation failure in critically ill adult patients. Crit Care 2009;13:233.
3. Lewis KE. Transtracheal lignocaine: Effective treatment for postextubation stridor. Anaesth Intensive Care 2007;35: 128-31.
4. Murphy GS, Szokol JW, Marymont JH, Greenberg SB, Avram MJ, Vender JS. Residual neuromuscular blockade and critical respiratory events in the postanesthesia care unit. Anesth Analg 2008;107:130-7.
5. Roberts KW, Crnkovic A, Steiniger JR. Post-anesthesia paradoxical vocal cord motion successfully treated with
midazolam. Anesthesiology 1998;89:517-9.
6. Ajithkumar AK, Botha JA. Metabolic stridor: A case report. Crit Care Resusc 2004;6:277-9.
7. Moralee SJ, Reilly PG. Metabolic stridor: Bilateral vocal cord abductor paralysis secondary to hypokalaemia? J Laryngol Otol 1992;106:56-7.
8. Laing CM, Unwin RJ. Renal tubular acidosis. J Nephrol 2006;19 Suppl 9:S46-52.
9. Emmett EK. Overview and Pathophysiology of Renal Tubular Acidosis and the Effect on Potassium Balance. Available from: https://www.uptodate.com/contents/overview-and-pathophysiology-of-renal-tubular-acidosis and the-effect-on-potassium-balance. [Last accessed on 2017 Mar 27].
10. Izzedine H, Launay-Vacher V, Isnard-Bagnis C, Deray G. Drug-induced Fanconi’s syndrome. Am J Kidney Dis 2003;41:292-309.
11. Hall AM, Bass P, Unwin RJ. Drug-induced renal Fanconi syndrome. QJM 2014;107:261-9.
| How to Cite this Article: Mallard CJ, Srour H. A metabolic cause of post-operative stridor in a patient with known mediastinal masses. Journal of Anaesthesia and Critical Care Case Reports May-Aug 2017;3(2):30-33. |